Adipogenic Differentiation in Research: Mechanisms, Cell Models and Experimental Strategies
Apr 17,2026
Adipogenic differentiation is a highly coordinated cellular process where precursor cells commit to the adipocyte lineage, typically induced in vitro using adipogenic differentiation medium that facilitates the initiation and progression of adipocyte differentiation. These cells accumulate triglyceride-rich lipid droplets and develop endocrine functions. This mechanism plays a crucial role in adipose tissue development, energy balance, and metabolic control. In vitro modeling of this process offers vital insights into physiological adipogenesis and its disruption in disease conditions. This article provides a systematic examination of the molecular regulation, cellular models, culture methods, validation approaches, and translational applications essential to modern adipogenesis research.
Table of Contents
1. Regulatory mechanisms and signaling pathways governing adipogenic differentiation
2. Comparing adipogenic potential and selecting cell models of mesenchymal and pre-adipocyte types
3. Essential components of adipogenic media
4. Experimental workflows and phenotypic validation
5. Translational relevance of adipogenic models in metabolic disease research
01 Regulatory mechanisms and signaling pathways governing adipogenic differentiation
Adipogenic differentiation is directed by a hierarchical transcriptional network initiated by extracellular cues. The master regulators peroxisome proliferator-activated receptor gamma (PPARγ) and CCAAT/enhancer-binding protein alpha (C/EBPα) form a positive feedback loop that activates adipocyte-specific genes including FABP4, ADIPOQ, and LPL[1]. Early commitment requires suppression of anti-adipogenic pathways; notably, canonical Wnt/β-catenin signaling maintains progenitor quiescence, and its downregulation is essential for differentiation initiation [2]. Pro-adipogenic signals include bone morphogenetic proteins (BMP4 and BMP7), which promote lineage specification through SMAD-dependent activation of PPARγ [3]. Insulin/IGF-1signaling via PI3K/Akt promotes the translocation of glucose transporter 4 (GLUT4) and the expression of lipogenic enzymes while glucocorticoids induce mitotic clonal expansion which is a prerequisite event for the cell cycle[4]. Recent epigenomic studies reveal dynamic DNA demethylation at the PPARγ2 promoter and H3K27ac enrichment at enhancer regions during differentiation [5]. Non-coding RNAs, particularly miR-27b and miR-130, fine-tune this cascade by targeting PPARγ mRNA [6]. Understanding these integrated pathways enables targeted manipulation of adipogenesis for research and therapeutic development.
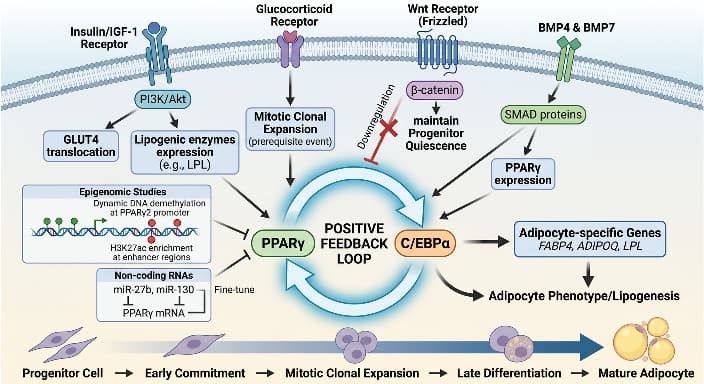
Fig. 1 Lipogenic-induced signal transduction.
02 Comparing adipogenic potential and selecting cell models of mesenchymal and pre-adipocyte types
Cell model selection profoundly impacts experimental outcomes in adipogenesis studies. Primary human mesenchymal stem cells (MSCs) derived from distinct tissues exhibit variable differentiation capacities. Bone marrow derived MSCs (BM-MSCs) demonstrate moderate adipogenic potential but are influenced by donor age, comorbidities, and passage number [7]. Adipose derived mesenchymal stem cells (AD-MSCs), isolated from the stromal vascular fraction of lipoaspirates, consistently show superior lipid accumulation and higher expression of adipogenic markers compared to BM-MSCs, attributed to their native adipose microenvironment [8]. Umbilical cord derived mesenchymal stem cells (UC-MSCs) offer ethical advantages and robust proliferation but generally require enhanced induction protocols due to inherently lower baseline adipogenic competence [9]. The murine 3T3-L1 preadipocyte cell line remains a gold standard for mechanistic studies of 3T3-L1 differentiation owing to its homogeneity, reproducibility, and well-characterized protocol; however, species-specific differences necessitate cautious translation to human physiology [10]. Critical considerations in mesenchymal stem cells culture include achieving >95% confluency prior to induction, using low-passage cells (< P6), and validating donor-specific variability. Table 1 provides a comparative overview to guide model selection.
Table 1. Comparative Attributes of Common Adipogenic Cell Models
| Cell Model | Differentiation Efficiency | Key Advantages | Primary Limitations |
| Bone marrow MSCs | Moderate | Multipotent, extensively characterized | Invasive harvest, donor variability |
| Adipose MSCs | High | Abundant source, high yield, robust lipid accumulation | Stromal heterogeneity, donor BMI influence |
| Umbilical cord MSCs | Low to moderate | Non-invasive collection, high proliferative capacity | Lower adipogenic baseline, batch variability |
| 3T3-L1 cells | High (standardized) | Reproducible, homogeneous, established protocols | Non-human origin, genetic drift risk |
03 Essential components of adipogenic media
The formulation of adipogenic media is pivotal for efficient differentiation. A foundational cocktail includes dexamethasone (0.5-1 μM; induces cell cycle exit and PPARγ expression), 3-isobutyl-1-methylxanthine (IBMX; 0.5 mM; elevates cAMP to activate PKA), insulin (1-10 μg/mL; stimulates glucose uptake and lipogenesis), and indomethacin (50-100 μM; PPARγ agonist) [11]. For human MSCs, rosiglitazone (1-2 μM) is frequently added to potentiate PPARγ activity [12]. The basal medium is typically high-glucose DMEM supplemented with 10% fetal bovine serum (FBS), L-glutamine, and antibiotics. Critical optimization parameters include serum batch validation (due to variable adipokine content), precise timing of induction (typically 72 hours), and transition to maintenance media containing insulin alone [13]. Species- and source-specific adjustments are essential: AD-MSCs often require reduced dexamethasone concentrations, while UC-MSCs may benefit from BMP4 supplementation [14]. Emerging trends favor xeno-free formulations using human platelet lysate or defined lipid supplements to enhance clinical translatability and reduce experimental variability [15].
04 Experimental workflows and phenotypic validation
A standardized workflow begins with expanding cells to 100% confluency in growth medium. Induction commences with adipogenic media for 3-7 days, followed by maintenance media (insulin-supplemented) for 7-14 days, with media refreshed every 48 hours [16]. Phenotypic validation employs multi-parametric approaches. Morphologically, the accumulation of intracellular lipids is measured through Oil Red O staining, a lysochrome dye that attaches to neutral lipids, producing red-orange droplets observable under brightfield microscopy.Staining intensity is quantified measured by spectrophotometry after dye elution (e.g., isopropanol at 510 nm) [17]. Molecular confirmation includes qPCR for PPARγ, C/EBPα, and FABP4, and Western blotting for adiponectin or perilipin. Functional assays assess insulin-stimulated glucose uptake or catecholamine-induced lipolysis. Rigorous controls include undifferentiated cells and vehicle-treated samples. Fig. 2 outlines the protocol workflow, while Fig. 3 illustrates representative Oil Red O outcomes.
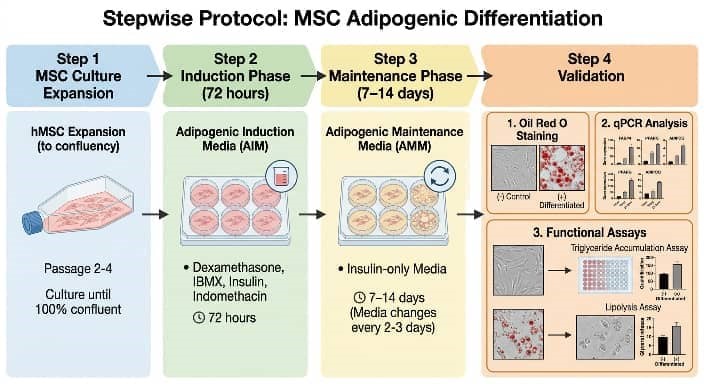
Fig. 2 Adipogenic Differentiation Workflow Diagram.
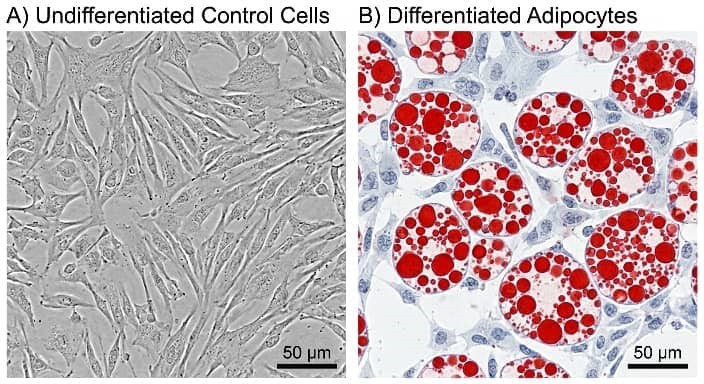
Fig. 3 Oil Red O Staining Validation.
05 Translational relevance of adipogenic models in metabolic disease research
Adipogenic models are indispensable for deciphering pathophysiology and developing interventions for metabolic disorders. Patient-derived AD-MSCs or iPSC-differentiated adipocytes model genetic lipodystrophies or insulin resistance syndromes, revealing cell-autonomous defects[18]. In obesity research, hypertrophic adipocytes generated from high-BMI donors exhibit elevated inflammatory cytokine secretion (e.g., IL-6, MCP-1), mimicking adipose tissue inflammation [19]. Drug screening platforms utilizing 3T3-L1 differentiation or human MSCs have identified novel PPARγ modulators with reduced side-effect profiles compared to classical thiazolidinediones [20]. Co-culture systems integrating adipocytes with hepatocytes or macrophages elucidate inter-organ crosstalk in metabolic syndrome [21]. CRISPR-Cas9-edited adipogenic models (e.g., FTO or IRS1 knockouts) dissect gene-specific roles in lipid metabolism [22]. Emerging 3D adipose organoids incorporating vascular cells enhance physiological relevance for studying adipose expansion and fibrosis [23]. These metabolic disease models accelerate therapeutic discovery while reducing reliance on animal studies, bridging molecular insights to clinical applications.
References
[1] Rosen ED, Spiegelman BM. Molecular regulation of adipogenesis. Annu Rev Cell Dev Biol. 2000; 16:145–171.
[2] Bennett CN, Longo KA, Wright WS, et al. Regulation of Wnt signaling during adipogenesis. J Biol Chem. 2002; 277(34):30998–31004.
[3] Tang QQ, Otto TC, Lane MD. Commitment of C3H10T1/2 pluripotent stem cells to the adipocyte lineage. Proc Natl Acad Sci USA. 2004; 101(26):9607–9611.
[4] Lefterova MI, Zhang Y, Steger DJ, et al. PPARγ and C/EBP factors orchestrate adipocyte biology. Genes Dev. 2008; 22(14):1851–1856.
[5] Siersbæk R, Nielsen R, Mandrup S. Transcriptional networks and chromatin remodeling controlling adipogenesis. Trends Endocrinol Metab. 2012; 23(2):56–64.
[6] Kim SY, Kim AY, Lee HW, et al. miR-27a is a negative regulator of adipocyte differentiation. Biochem Biophys Res Commun. 2010; 392(3):323–328.
[7] Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001; 7(2):211–228.
[8] Gimble JM, Katz AJ, Bunnell BA. Adipose-derived stem cells for regenerative medicine. Circ Res. 2007; 100(9):1249–1260.
[9] Wang L, Ott L, Seshareddy K, et al. Comparison of human umbilical cord matrix stem cells and adipose tissue-derived stem cells. Stem Cells Dev. 2011; 20(5):877–886.
[10] Gregoire FM, Smas CM, Sul HS. Understanding adipocyte differentiation. Physiol Rev. 1998; 78(3):783–809.
[11] Ramírez-Zacarías JL, Castro-Muñozledo F, Kuri-Harcuch W. Quantitative determination of adipose conversion by Oil Red O staining. Histochemistry. 1992; 97(3):221–226.
[12] Farmer SR. Transcriptional control of adipocyte formation. Cell Metab. 2006; 4(4):263–273.
[13] Planat-Benard V, Silvestre JS, Cousin B, et al. Plasticity of human adipose lineage cells toward endothelial cells. Circulation. 2004; 109(5):656–663.
[14] Foubert P, Barra JG, Monroe WT. Rapid endothelialization of tissue engineered vascular grafts. Biomaterials. 2010; 31(22):5831–5837.
[15] Sensebé L, Gadelorge M, Fleury-Cappellesso S. Production of mesenchymal stromal/stem cells according to good manufacturing practices. Stem Cells Int. 2015; 2015:1–10.
[16] Green H, Kehinde O. An established preadipose cell line and its differentiation in culture. Cell. 1974; 3(1):127–139.
[17] Koopman R, van Baak MA, Håkansson J, et al. Quantification of Oil Red O staining in adipocytes. Anal Biochem. 2001; 292(1):139–142.
[18] Garg A. Clinical review: Lipodystrophies: genetic and acquired body fat disorders. J Clin Endocrinol Metab. 2011; 96(11):3313–3325.
[19] Lumeng CN, Bodzin JL, Saltiel AR. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin Invest. 2007; 117(1):175–184.
[20] Choi JH, Banks AS, Kamenecka TM, et al. Antidiabetic actions of a non-agonist PPARγ ligand blocking Cdk5-mediated phosphorylation. Nature. 2011; 477(7365):477–481.
[21] Kanda H, Tateya S, Tamori Y, et al. MCP-1 contributes to macrophage infiltration into adipose tissue. J Clin Invest. 2006; 116(6):1494–1505.
[22] Claussnitzer M, Dankel SN, Kim KH, et al. FTO obesity variant circuitry and adipocyte browning in humans. N Engl J Med. 2015; 373(10):895–907.
[23] Lee J, Choi J, Lee H, et al. 3D adipose tissue models for metabolic disease research. Adv Healthc Mater. 2020; 9(15):e2000351.
Prev: World Parkinson’s Day: Deciphering Disease Mechanisms Through Cellular Models


