Primary Cell Immortalization: Protocols for Culture, Cryopreservation, and Resuscitation
Apr 29,2026
Immortalized primary cells retain key characteristics of primary cells while allowing sustained proliferation, making them essential in cell biology, pharmacology, and related fields. However, suboptimal handling during culture, cryopreservation, or recovery can reduce viability, impair adhesion, and increase contamination risk, ultimately compromising experimental outcomes.
In this issue of Cell Culture Academy, we outline standardized protocols for culture, cryopreservation, and recovery to improve cell culture success rates.
I. Cell Culture
Culturing immortalized primary cells requires mimicking the in vivo environment with appropriate nutrients and growth conditions. Key steps include seeding, medium change, and passaging.
1. Cell Seeding
Proper seeding density and uniform distribution are critical for efficient attachment and stable growth.
Procedure
Centrifuge cells (post-thaw or post-passage) at 1,000 rpm for 5 min and discard the supernatant.
Resuspend in prewarmed (37℃) complete medium to obtain a single-cell suspension.
Add medium to the culture flask, then seed cells at the desired density (typically 1 × 105-1 × 106 cells/mL) by adding the calculated volume of cell suspension along the flask wall and mixing gently.
Adjust to the recommended final volume with complete medium.
Gently rock to distribute cells evenly and prevent aggregation.
Incubate at 37℃ with 5% CO2 for 24-48 h, minimizing disturbance to allow proper attachment.
2. Medium Change
During culture, nutrients are depleted and metabolic waste accumulates; therefore, change the medium every 2-3 days.
Procedure
In a biosafety cabinet, gently tilt the flask to pool the medium on one side.
Aspirate the spent medium without disturbing adherent cells.
Rinse with prewarmed (37℃) PBS, gently rock, and aspirate (repeat if needed during the first change).
Add fresh prewarmed complete medium and return to the incubator.
3. Cell Passaging
Passage cells at 80-90% confluence.
Procedure
Aspirate the medium and rinse twice with PBS to remove residual serum, which inhibits enzymatic activity.
Add dissociation enzyme to just cover the surface and incubate at 37℃ for 1-3 min.
Monitor microscopically; once cells round up and begin to detach, proceed according to the enzyme used:
Trypsin: Immediately add 3-5 volumes of serum-containing medium or a trypsin inhibitor to neutralize.
Mild dissociation enzymes (e.g., Accutase): No neutralization required.
Gently pipette to obtain a single-cell suspension.
Centrifuge at 1,000 rpm for 5 min, discard the supernatant, and resuspend in fresh medium.
Seed into new flasks at the recommended split ratio (typically 1:2-1:4) and continue culture.
Notes
Limit enzymatic digestion time to prevent cell damage.
Pipette gently to minimize shear stress.
For poorly adherent cells, pre-coat culture surfaces (e.g., glass coverslips, plates, or confocal dishes) with extracellular matrix components such as collagen or poly-L-lysine to improve attachment.
Ⅱ. Cell Cryopreservation
Cryopreservation enables long-term cell storage. Use healthy cells at 80-90% confluence.
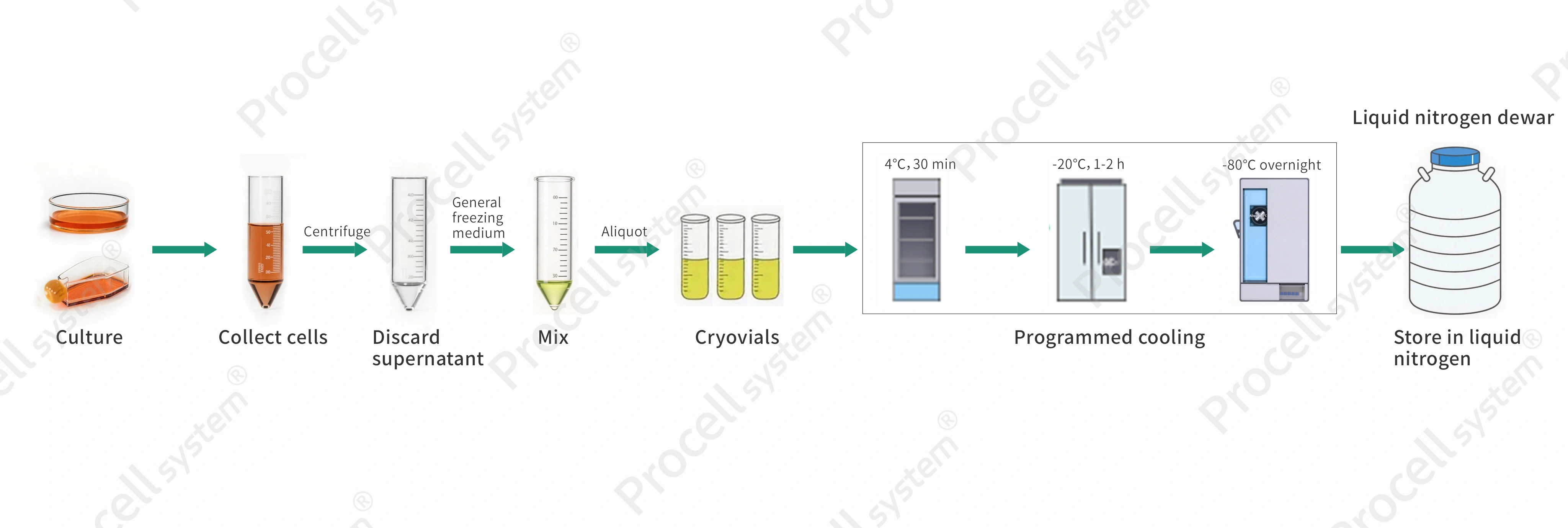
Figure 1. Illustration of programmable freezing operation
Procedure
Replace the culture medium within 24 h before freezing.
Harvest cells and prepare a single-cell suspension with >90% viability.
Adherent cells: Detach and collect by centrifugation using standard passaging methods.
Suspension cells: Collect directly by centrifugation.
Centrifuge at 1,000 rpm for 5 min and discard the supernatant.
Resuspend the pellet in pre-chilled cryopreservation medium and gently pipette to mix, adjusting to 2×106-5 × 106 cells/mL.
Recommended Product: General Freezing Medium
Aliquot 0.5-1 mL per cryovial, secure caps, and label (e.g., cell type, date).
Freeze using a stepwise cooling program (4℃ for 30 min; -20℃ for 1-2 h; -80℃ overnight).
For long-term storage, transfer to liquid nitrogen after > 16 h at -80℃.
Notes
Conduct a pilot cryopreservation test at least one week before formal freezing.
Use a controlled-rate freezing container to ensure gradual cooling and improve post-thaw viability.
III. Cell Resuscitation
Cell resuscitation reverses cryopreservation; rapid thawing minimizes cellular damage.
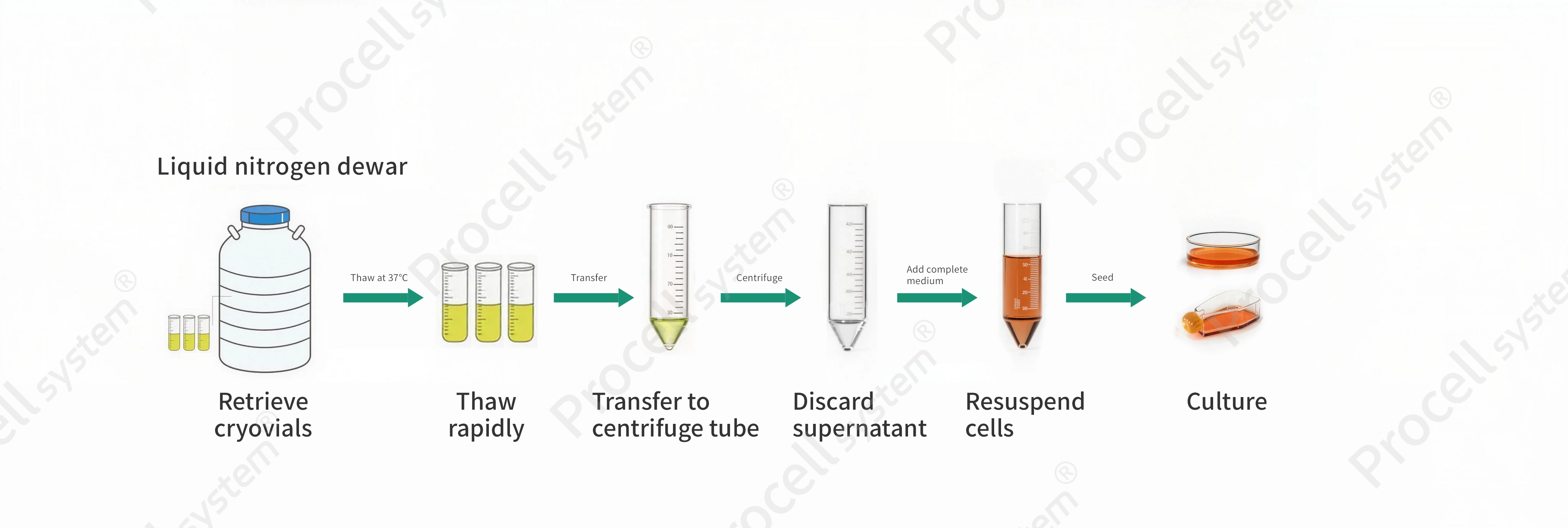
Figure 2. Illustration of cell resuscitation operation
Procedure
Pre-warm complete medium in a 37℃ water bath and prepare culture flasks and centrifuge tubes.
Remove the cryovial from liquid nitrogen, place it in a protective glove or sleeve, and immediately immerse it in a 37℃ water bath.
Gently agitate until fully thawed (1-2 min).
Transfer the cell suspension to a centrifuge tube containing 3-5 volumes of pre-warmed medium.
Mix gently and centrifuge at 1,000 rpm for 5 min, then discard the supernatant. Resuspend the pellet in complete medium, seed into a culture flask, and adjust to the desired volume.
Incubate under standard conditions.
Notes
Post-thaw cells are fragile; use a slightly higher seeding density. Do not disturb cultures for at least 24 h to allow attachment. If viability is low, increasing serum concentration may aid recovery.
IV. Common Issues and Troubleshooting
1. Poor attachment or low viability
Confirm that the culture medium and serum concentration are appropriate. Minimize digestion time and handle cells gently during pipetting. Thaw rapidly and maintain stable incubator conditions (CO2 concentration and temperature).
2. Slow growth after passaging
Often due to an overly low split ratio; increase seeding density as needed. Ensure the medium is fresh and properly stored. Immortalized cells, especially human-derived cells, are at higher risk of mycoplasma contamination; perform routine testing.
3. Contamination
If the medium appears turbid or contains flocculent material or black particles, discard the culture immediately. Disinfect the workspace and equipment thoroughly (e.g., UV sterilization of biosafety cabinets, re-sterilization of consumables) to prevent cross-contamination. Maintain strict aseptic technique.
Although culture, cryopreservation, and thawing may seem straightforward, each step impacts cell quality. Consistent aseptic technique, gentle handling, and stable culture conditions are critical for maintaining viability.
Prev: Overcoming Replicative Limits: Mechanisms and Applications of Primary Cell Immortalization


