Streamlining Primary Cell Research with Specialized Isolation Kits for Higher Purity and Reproducibility
Mar 06,2026
Primary cells, directly isolated from living tissues, are the gold standard for in vitro research models that recapitulate in vivo conditions. Unlike immortalized cell lines, they maintain native tissue characteristics, making them essential for drug discovery, disease modeling, toxicology screening, and regenerative medicine. However, the scientific value of primary cell research depends on the quality of the initial isolation, a technically demanding process with consistency and reproducibility issues. Commercial primary cell isolation kits have revolutionized this step by providing standardized, optimized protocols for high purity, viable cells. These kits tackle tissue specific issues with ready enzyme blends, safeguarding buffers, and streamlined procedures.
They have democratized primary cell research, improving data quality and experimental reproducibility. This review examines how specialized isolation kits streamline primary cell research across disciplines, focusing on challenging cell types. It delves into isolation obstacles, the way contemporary kits surmount them, post isolation cultural practices, and problem solving approaches. Standardization through these kits results in improved research outcomes and accelerated scientific breakthroughs.
Table of Contents
1. What is a primary cell isolation kit used for?
2. Benefits of using primary cells in research
3. Challenges in primary cell isolation
4. How do primary cell isolation kits improve success rates?
5. Protocols for primary cell culture after isolation
6.Troubleshooting common issues with primary cell isolation
01 What is a primary cell isolation kit used for?
A primary cell isolation kit represents a meticulously engineered system designed to dissociate complex tissue structures into viable, functional single cells while preserving their native morphology, surface markers, and physiological properties. These integrated solutions typically contain precisely formulated enzyme blends (including collagenase, dispase, trypsin, hyaluronidase, and DNase), specialized buffers with optimized pH and osmolarity, inhibitors to protect cellular integrity, and detailed, step by step protocols tailored to specific tissue types such as brain, liver, pancreas, adipose tissue, or vasculature[1].
The purpose of these kits is not just tissue dissociation; they lay the foundation for primary cell culture models that maintain in vivo like behavior in experiments. Unlike traditional laboratory prepared digestion solutions that need much optimization and give inconsistent results, modern commercial kits integrate years of research and refinement into standardized workflows. This standardization allows researchers of different expertise levels to get reproducible results regardless of their institution or background.
Primary cell isolation kits serve multiple critical functions in the research pipeline:
● Facilitating rapid tissue processing with minimized cell death.
● Preserving cell surface receptor integrity for downstream functional assays.
● Maximizing yield of target cell populations through selective dissociation.
● Reducing contamination from unwanted cell types.
● Standardizing procedures across different operators and laboratory environments.
● Decreasing overall processing time from tissue harvest to culture establishment.
The sophisticated design of modern isolation kits reflects a nuanced understanding of tissue microarchitecture. For instance, neural isolation kits employ gentle protease combinations that selectively digest connective tissue elements while preserving delicate neuronal processes and synaptic structures. Cardiac isolation kits contain collagenase blends specifically formulated to dissociate the dense collagen networks of heart tissue without damaging cardiomyocyte contractile apparatus.
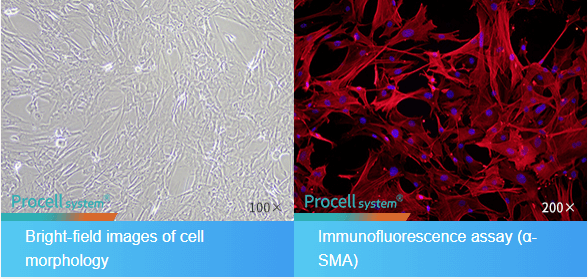
Fig. 1 Morphological and Immunofluorescence Identification of Mouse Brain Microvascular Pericytes.
This targeted approach represents a significant advancement compared to historical "one size fits all" digestion protocols, which often yielded suboptimal results. Specialized kits preserve cellular functionality instead of just achieving dissociation, enabling downstream applications that were previously difficult or impossible with conventionally isolated cells.
02 Benefits of using primary cells in research
Primary cells offer unparalleled biological relevance compared to immortalized cell lines, providing researchers with experimental models that faithfully replicate in vivo physiology, metabolism, and response patterns. This superior fidelity stems from their direct derivation from living tissues without genetic manipulation or extended culture adaptation, preserving the full complement of native cellular machinery, signaling pathways, and epigenetic regulation mechanisms that define cell behavior in living organisms.
The biological authenticity of primary cells is crucial for research validity across multiple dimensions. Genetically, they keep the complete diploid genome with intact regulatory regions, avoiding mutations, chromosomal rearrangements, and gene copy number variations seen in immortalized lines after multiple passages. Metabolically, they preserve tissue specific metabolic programs, including enzyme expression, substrate preferences, and energy production pathways, which influence drug responses and disease mechanisms. Phenotypically, they retain characteristic morphology, polarization, cell to cell communication, and functional outputs essential for modeling complex biological processes[2].
Table 1. Comparative Advantages of Primary Cells Versus Common Cell Lines for Specific Research Applications
| Research Application | Primary Cell Advantage | Cell Line Limitation |
| Drug Metabolism Studies | Express full complement of phase I/II metabolizing enzymes (CYP450s) at physiological levels | Often lack or have altered drug metabolism enzyme expression |
| Neuroscience Research | Maintain authentic neuronal networks, synaptic plasticity, and glial interactions | Lack complex network formation and physiological electrophysiological properties |
| Immunological Studies | Preserve native immune receptor diversity and signaling cascades | May exhibit aberrant immune responses due to transformation |
| Cancer Microenvironment | Retain patient specific tumor stromal interactions and heterogeneity | Absence of authentic tumor microenvironment components |
| Toxicology Screening | Express tissue specific toxicity pathways and detoxification mechanisms | Frequently show altered sensitivity to toxins due to adaptive mutations |
Primary neuronal cells show advantages in neuroscience research. Unlike neuroblastoma cell lines with simplified electrophysiology, primary neurons maintain mature ion channel expression, form functional synapses with neurotransmitter release machinery, and display activity dependent plasticity for studying learning, memory, and neurodegenerative processes[3]. When co cultured with primary astrocytes isolated by specialized kits, these neuronal cultures incorporate glial regulation of synaptic function, neurovascular coupling, and metabolic support, which are absent in transformed cell models.
Similarly, primary astrocytes preserve critical in vivo functions such as glutamate uptake regulation, potassium buffering, blood brain barrier maintenance, and inflammatory response modulation. Their ability to form gap junction coupled networks and respond to physiological calcium signaling enables researchers to study neurological disorders from a systems view rather than isolated cellular pathways. This holistic approach is particularly valuable in modeling complex conditions like Alzheimer's disease, where astrocyte dysfunction contributes significantly to disease progression through multiple interconnected mechanisms.
Primary fibroblasts are a crucial research model as biological fidelity affects experimental outcomes. Unlike immortalized fibroblast lines, they retain donor specific responses to growth factors etc., which vary among individuals due to age, genetics and disease state. This variation makes them valuable for studying wound healing, fibrotic disorders, aging and personalized medicine. For example, fibroblasts from scleroderma patients show distinct profibrotic signaling and extracellular matrix production that standard cell lines can't replicate.
The clinical relevance of primary cells extends to preclinical drug development, where their predictive value significantly outperforms traditional cell line models. In hepatotoxicity screening, primary human hepatocytes detect compound induced liver injury with approximately 70-80% accuracy compared to clinical outcomes, while common hepatic cell lines achieve only 30-40% predictive correlation. Similarly, in cardiac safety pharmacology, primary cardiomyocytes identify arrhythmogenic compounds through physiologically relevant electrophysiological responses that immortalized lines frequently miss due to altered ion channel expression[5].
Although these striking benefits exist, technical difficulties in steady isolation and culture have traditionally restricted the complete potential of primary cell research. It is exactly at this point that specialized isolation kits have had their most substantial impact by standardizing the preliminary cell collection procedure, these kits guarantee that the innate biological advantages of primary cells are maintained across experimental processes, turning cellular genuineness into research dependability.
03 Challenges in primary cell isolation
Despite their unparalleled biological relevance, primary cell isolation remains a technically demanding process fraught with challenges that can compromise experimental outcomes if not properly addressed. These difficulties stem from fundamental biological complexities: tissues vary dramatically in structural composition, cellular density, extracellular matrix architecture, and sensitivity to environmental perturbations. These inherent variations demand specialized approaches rather than universal protocols, creating significant barriers for researchers without extensive experience in primary cell techniques.
Tissue heterogeneity is the most fundamental challenge in primary cell isolation. Neural tissue shows this complexity as it's soft and sensitive to mechanical and enzymatic stress, with multiple cell types in a delicate extracellular matrix. The dissociation process must balance tissue breakdown to free cells and preserve fragile neuronal processes and synaptic structures, which can be damaged by mild enzymatic overexposure. Similarly, liver tissue has difficulties; its dense, vascularized architecture resists standard enzymatic digestion, yet its highly metabolic hepatocytes are vulnerable to hypoxia and oxidative stress during long term processing. Traditional cell isolation methodologies, from decades old protocols, have critical limitations that affect reproducibility and cell quality.
● Extreme sensitivity to minor procedural variations: Temperature fluctuations of even 1-2°C, slight differences in enzyme concentration, or minute variations in incubation duration can dramatically alter outcomes
● Inadequate protection against cellular stress: Standard buffers frequently lack antioxidants, energy substrates, or apoptosis inhibitors needed to maintain viability during the metabolically demanding dissociation process.
● Poor cell type specificity: Conventional enzyme blends non selectively digest all tissue components, yielding heterogeneous cell mixtures. These mixtures need additional purification steps, which further reduce recovery.
● Batch to batch variability: Laboratory prepared enzyme solutions exhibit significant activity differences between preparations due to source variations and storage conditions.
● Incomplete dissociation: Dense tissues frequently yield multicellular aggregates that compromise downstream applications like flow cytometry, single cell RNA sequencing, or precise cell counting.
Table 2. Tissue Specific Challenges in Primary Cell Isolation and Traditional Limitations
| Tissue Type | Structural Characteristics | Primary Isolation Challenges |
| Brain/CNS | Soft parenchyma with dense neural networks | Maintaining neuronal viability and process integrity; separating neuronal/glial populations |
| Liver | Highly vascularized with dense collagen networks | Hepatocyte fragility; sinusoidal endothelial cell contamination; hypoxic damage |
| Skin/Dermis | Stratified epithelium over dense collagen matrix | Keratinocyte fibroblast separation; maintaining stem cell populations |
| Adipose | Lipid rich cells within fibrovascular stroma | Adipocyte fragility; stromal vascular fraction contamination |
| Cardiac | Highly organized contractile tissue with extensive vasculature | Cardiomyocyte sensitivity to calcium; fibroblast overgrowth |
The consequences of suboptimal isolation extend far beyond simple yield reduction. Compromised cells often exhibit:
● Altered gene expression profiles due to stress response activation during harsh dissociation.
● Reduced functional capacity such as diminished electrophysiological activity in neurons or decreased albumin production in hepatocytes.
● Accelerated senescence leading to shortened experimental windows.
● Unintended phenotypic drift where cells adapt to culture conditions rather than maintaining native characteristics.
● Batch dependent experimental variability that obscures biological effects and reduces statistical power.
These challenges are especially acute in neuroscience research, where primary neuronal isolation demands high precision. Neurons are in complex 3D networks connected by delicate axons and dendrites, which are easily sheared during mechanical dissociation. Their high metabolic rate and dependence on astrocyte derived factors make them very vulnerable to isolation stress. Conventional trypsin based protocols mostly yield glial cells with little neuronal recovery, and overaggressive mechanical trituration destroys synaptic structures crucial for functional studies.
Similarly, primary astrocyte isolation faces the dual challenge of achieving sufficient yield while preventing microglial and oligodendrocyte precursor contamination. Many traditional protocols yield "astrocyte" cultures containing 30-40% non astrocytic cells that significantly alter experimental outcomes in inflammation and neuroprotection studies. The subsequent need for additional purification steps (such as shake off procedures or immunopanning) introduces further stress and reduces overall recovery.
Primary fibroblast isolation, while technically less demanding than neural cells, still faces significant hurdles. Dermal fibroblasts exist embedded within a dense collagen matrix that requires aggressive enzymatic treatment, yet excessive collagenase exposure damages surface receptors critical for migration and signaling studies. Furthermore, fibroblast cultures frequently become contaminated with keratinocytes, endothelial cells, or immune cells that alter proliferation rates and cytokine responses. These contaminants may remain undetected without rigorous characterization but can substantially impact experimental reproducibility.
The technical expertise needed to overcome these challenges has historically been a significant barrier to primary cell research. Successful isolation often relied on tacit knowledge shared among lab members, such as subtle techniques for tissue mincing, trituration pressure, and visual cues for digestion endpoint, which were rarely documented in protocols. This expertise dependency led to the well known "lab to lab variability", undermining confidence in primary cell data and slowing the adoption of these superior biological models.
Additional practical constraints further complicated traditional isolation approaches:
● Time sensitivity: Many primary cells rapidly deteriorate after tissue harvest, requiring immediate processing that conflicts with practical laboratory scheduling.
● Equipment requirements: Specialized tools like tissue choppers, perfusion systems, or gradient centrifugation equipment were often necessary but unavailable to many researchers
● Regulatory compliance: Increasing restrictions on biological materials handling demanded documentation and quality control often absent in traditional protocols.
● Scalability issues: Methods optimized for small tissue samples frequently failed when scaled to larger quantities needed for high throughput applications.
These multifaceted challenges underscore why primary cell research remained the domain of specialized laboratories despite the clear biological advantages of these models. The development of specialized isolation kits specifically addressed these limitations through standardized formulations, optimized protocols, and protective components that collectively transform this historically challenging procedure into a reliable, accessible technique for researchers across diverse disciplines.
04 How do primary cell isolation kits improve success rates?
Commercial primary cell isolation kits address historical primary cell research challenges via scientifically engineered solutions that standardize and optimize the dissociation process. They achieve superior outcomes through sophisticated biochemical formulations based on knowledge of tissue microarchitecture, cellular stress responses, and enzyme kinetics, not just convenient reagent packaging. By standardizing the procedure, they improve success rates and lower the technical expertise barrier.
The core innovation of modern isolation kits is their precisely balanced enzyme combinations for different tissue types. Unlike traditional single enzyme methods, advanced kits use multi enzyme
cocktails. For example, in neural tissues, gentle protease blends with low concentration papain or
accutase degrade connective tissue proteins while preserving neuronal structures. These
formulations also include protease inhibitors to halt enzymatic activity and prevent cellular damag, a level of control not possible with conventional methods.
Liver dissociation kits exemplify this precision engineering through collagenase Type IV formulations specifically designed to digest the unique collagen types (I, III, IV) present in hepatic tissue while preserving hepatocyte polarity and metabolic function.
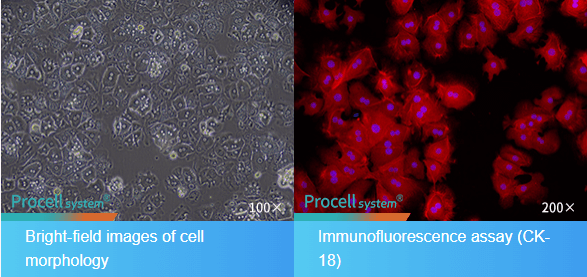
Fig. 2 Morphological and CK-18 Immunofluorescence Identification of Mouse Hepatic Parenchymal Cells.
The impact of these specialized formulations is reflected in dramatic improvements in key performance metrics compared to traditional methods:
● Viability increases from typical ranges of 60-70% with conventional methods to 85-95% with optimized kits.
● Yield improvements of 2-3 fold for difficult isolate cell types like neurons and hepatocytes.
● Functional preservation demonstrated by higher metabolic activity, electrophysiological responsiveness, and receptor expression.
● Reduced processing time from several hours to 30-60 minutes for most tissue types.
● Enhanced purity with target cell populations reaching >90% without additional purification steps.
The standardization of commercial kits addresses the most persistent challenge in primary cell research: reproducibility. Each kit lot undergoes strict quality control to ensure consistent enzyme activity, buffer composition, and performance, eliminating batch to batch variability in laboratory prepared solutions. For multi center or longitudinal studies, this consistency ensures observed biological effects reflect experimental variables. Moreover, the kits’ optimized protocols reduce. The accessibility of specialized kits benefits not only technical performance but also practical laboratory work. Detailed protocols with timing recommendations, visual cues, and troubleshooting guides help less experienced researchers achieve professional results. Many kits now offer digital resources like videos, webinars, and mobile apps to guide users through complex procedures.
The economic impact of specialized isolation kits is frequently underestimated. While kit costs may appear higher than raw enzyme purchases, comprehensive analysis reveals substantial overall savings through:
● Reduced technician time (often 50-70% less hands on processing time).
● Decreased repeat experiments due to failed isolations.
● Improved data quality leading to more conclusive results with fewer replicates.
● Extended experimental windows from healthier, longer lived cultures.
● Reduced waste disposal costs from fewer failed attempts.
These advancements have fundamentally transformed primary cell research feasibility. Previously, primary cell work needed dedicated core facilities or specialized expertise, but modern kits allow individual laboratories to regularly include these superior biological models in their research. This democratization has sped up discovery in multiple fields. In neuroscience, functional neuronal networks model complex disorders. In oncology, patient derived primary cells enable personalized drug screening. In regenerative medicine, consistent isolation protocols support cell therapy manufacturing.
05 Protocols for primary cell culture after isolation
Successful primary cell culture starts right after isolation, with attention to plating conditions, media composition, and environmental parameters for cell health and experimental utility. The transition from isolated cells to established cultures is a vulnerable period; improper handling can harm research outcomes despite good isolation. Modern isolation kits often include post isolation culture recommendations for standardization.
The post isolation phase needs focus on several key parameters. Plate primary cells within 30 minutes after isolation to reduce anoikis. Resuspend cells in pre warmed, tissue specific media. Plating density is crucial; optimal densities vary by cell type, e.g., 50,000-100,000 cells/cm² for astrocytes and 150,000-300,000 cells/cm² for neurons.
Substrate preparation is vital, especially for anchorage dependent cells. Neurons need adhesion molecules like poly L-lysine or laminin. Astrocytes adhere well to standard plastic and better on collagen coated surfaces. Fibroblasts attach to uncoated plastic and benefit from fibronectin for some studies.
Media formulation is critical. Modern approaches favor defined, serum free media over high serum ones for better batch consistency and control. These formulations contain specific combinations of substances to maintain cell differentiation.
Primary neuronal cultures need special handling. Add neurotrophic factors after plating and mitotic inhibitors after 48-72 hours. Do half media changes every 3-4 days. Successful cultures show neurite outgrowth and electrical activity by day 7-10.
Primary astrocyte cultures from whole brain tissue benefit from purification by orbital shaking. Keep cultures at subconfluent densities for functional studies and change media every 2-3 days.
Primary fibroblast culture is relatively straightforward. Serum concentration affects behavior. For wound healing studies, reach 90-95% confluence before creating scratch wounds. Perform regular quality control checks.
Environmental control beyond CO₂ incubation matters. Temperature, humidity, and vibration control impact cell health. Specialized incubators with precise O₂ control can improve survival of sensitive cells.
Integrating post isolation protocols with high quality isolation kits creates a seamless workflow. This standardization makes primary cell research more reliable and accessible, enhancing data quality and reproducibility and accelerating discovery.
06 Troubleshooting common issues with primary cell isolation
Even with optimized commercial kits, primary cell isolation may face technical challenges that need systematic troubleshooting. These issues usually show as reduced viability, low yield, contamination, or functional impairment in subsequent cultures. Developing a diagnostic framework helps researchers identify root causes quickly and take corrective measures, minimizing wasted samples and delays. This section offers a comprehensive troubleshooting guide by symptom, with solutions validated by practical experience.
Low cell yield is a frequent isolation challenge with multiple potential causes. When yield is below expectations, researchers should evaluate each isolation step. Delayed tissue processing is a common cause; primary cells deteriorate after harvest, with significant viability loss in 1-2 hours for highly metabolic tissues. Implementing strict time protocols from harvest to dissociation (ideally <60 minutes) improves outcomes. For labs receiving external tissue, specialized transport media can extend the window to 4-6 hours without significant quality loss.
Table 3. Systematic Troubleshooting Guide for Common Primary Cell Isolation Problems
| Symptom | Likely Causes | Diagnostic Tests | Recommended Solutions | Prevention Strategies |
| Low Cell Yield | Delayed processing (>2h post harvest); Incomplete tissue dissociation; Cell loss during washing steps; Excessive cell death during isolation | Compare yield to tissue weight benchmarks; Check dissociation completeness microscopically; Assess viability at multiple timepoints | Initiate processing immediately; Increase enzyme concentration 10-20%; Add additional dissociation step; Reduce centrifugation force/speed | Maintain a strict timeline from harvest to processing, pre chill all equipment, and use wide bore pipettes for all transfers |
| Poor Viability | Enzyme overexposure; Mechanical stress during trituration; Temperature fluctuations; Endotoxin contamination; Hypoxic conditions during processing | Time course viability testing; Compare different trituration techniques; Measure endotoxin levels in reagents | Reduce enzyme incubation time 25-50%; Use fire polished pipettes with wide bores; Maintain strict temperature control; Filter sterilize all buffers; Process tissues in oxygen permeable containers | Titrate enzymes for each tissue batch; Pre warm all solutions to exact temperatures; Use certified low endotoxin reagents |
| Fibroblast Contamination in Neural Cultures | Incomplete microglial/oligodendrocyte removal; Fibroblast migration from meninges; Insufficient antimitotic treatment | Immunostaining for fibronectin and GFAP; Time lapse imaging of contaminant growth patterns | Extended orbital shaking (18-24h at 250rpm); Antimitotic treatment with Ara C (2-5μM); Meningeal removal prior to dissociation | Careful meningeal stripping under microscope; Differential adhesion purification; Magnetic sorting for neural markers |
| Red Blood Cell Contamination | Inadequate perfusion; Fragile vascular tissues; Insufficient RBC lysis | Visual inspection of cell pellet color; Flow cytometry for CD235a+ cells | Hypotonic lysis buffer treatment (30-60s); Density gradient centrifugation; Magnetic CD235a depletion | Perfuse tissues thoroughly before harvest; Use gentle dissociation for vascular tissues; Include RBC lysis buffer in protocol |
| Poor Neurite Outgrowth | Suboptimal coating quality; Insufficient neurotrophic support; Glial overgrowth; Contaminating proteases | Test coating efficiency with fluorescent beads; Measure BDNF/GDNF levels; Assess glial:neuron ratio | Re coat plates with fresh poly-L-lysine/laminin; Supplement with additional BDNF (50ng/mL) and GDNF (50ng/mL); Add Ara C earlier (24h post plating) | Verify coating solution pH and concentration; Use freshly prepared growth factors; Filter sterilize all supplements |
Poor cell viability (<70%) after isolation often indicates excessive stress during dissociation. Enzymatic overexposure, due to many tissues having a narrow dissociation window, is the main cause. Time course testing (dividing samples and stopping reactions every 5 minutes) can solve this. Mechanical stress during trituration, especially for fragile cells, is also an issue; using fire polished glass pipettes reduces shear forces. For mammalian tissues, dissociation temperature should be precisely 37°C.
Fibroblast contamination in neural cultures, often from incomplete meningeal removal, can be addressed by microscopic meningeal stripping before dissociation. Differential adhesion and pharmacological approaches can handle residual fibroblasts.
RBC contamination in isolations from vascularized tissues is common. Density gradient centrifugation or brief hypotonic lysis can remove RBCs, and thorough tissue perfusion before harvest is the best prevention.
Poor cell attachment may result from suboptimal coating or media. For neurons, poly L lysine coating has specific requirements, laminin should be added before plating, and neuronal plating media should be serum free.
Unexpected cellular changes suggest underlying problems. Regular immunophenotyping for lineage specific markers can identify issues before they affect data.
Kit component storage and handling impact isolation success. Enzyme solutions should be aliquoted and stored at 80°C, and buffers with calcium or magnesium should be filter sterilized. A kit inventory log helps with proactive replacement.
The most effective troubleshooting is prevention. Keeping detailed records, standardizing operator technique, and implementing quality control checkpoints can avoid problems.
Developing a systematic troubleshooting approach turns challenges into opportunities for protocol improvement, leading to more reliable primary cell research.
References:
[1]Freshney RI. Culture of Animal Cells: A Manual of Basic Technique and Specialized Applications. 7th ed. Hoboken, NJ: Wiley Blackwell; 2016.
[2]Li H, Zhang Y. Primary cell culture: A critical tool for biomedical research. Cell Biol Int. 2022;46(5):771 782. doi:10.1002/cbin.11768
[3]Schildge S, Bohrer C, Beck K, Schachtrup C. Isolation and culture of mouse cortical astrocytes. J Vis Exp. 2013;(71):e50079. doi:10.3791/50079
[4]Stoker MGP, Sheehan DJ, Bradshaw JR. Growth factor requirements of normal human fibroblasts in culture. J Cell Sci. 1985;77(1):1 11. doi:10.1242/jcs.77.1.1
[5]Gstraunthaler G, Lindl T, Romer J. Alternatives to the use of fetal bovine serum: serum free cell culture. ALTEX. 2013;30(4):389 402. doi:10.14573/altex.2013.4.389
[6]Bhatia SN, Yarmush ML, Toner M. Controlling cell interactions on micropatterned substrates. Biotechnol Prog. 1996;12(5):615 620. doi:10.1021/bp960062
Prev: How to Boost mRNA Transfection Efficiency: Key Strategies Explained
Next: Eliminate Cell Contamination: Key Types and Prevention Strategies at a Glance


