How to Boost mRNA Transfection Efficiency: Key Strategies Explained
Feb 25,2026
mRNA technology is transforming biomedical research. Owing to its expression without the need for nuclear entry and favorable safety profile, it plays an increasingly important role in gene function studies, vaccine development, and cell therapy. However, achieving high transfection efficiency while preserving cell viability and experimental reproducibility remains a common challenge.
In this issue of Cell Culture Academy, we systematically dissect the mRNA transfection workflow and present practical optimization strategies grounded in both theory and application.
Ⅰ. What Is mRNA Transfection?
mRNA expression is characterized by rapid onset, transient duration, and precise experimental control, minimizing cytotoxicity associated with sustained expression. These features make mRNA well suited for transient applications, including gain-of-function studies, CRISPR-Cas9-mediated genome editing, and rapid recombinant protein production.
However, this streamlined intracellular route also imposes stricter requirements on mRNA stability, delivery efficiency, and intracellular compatibility.
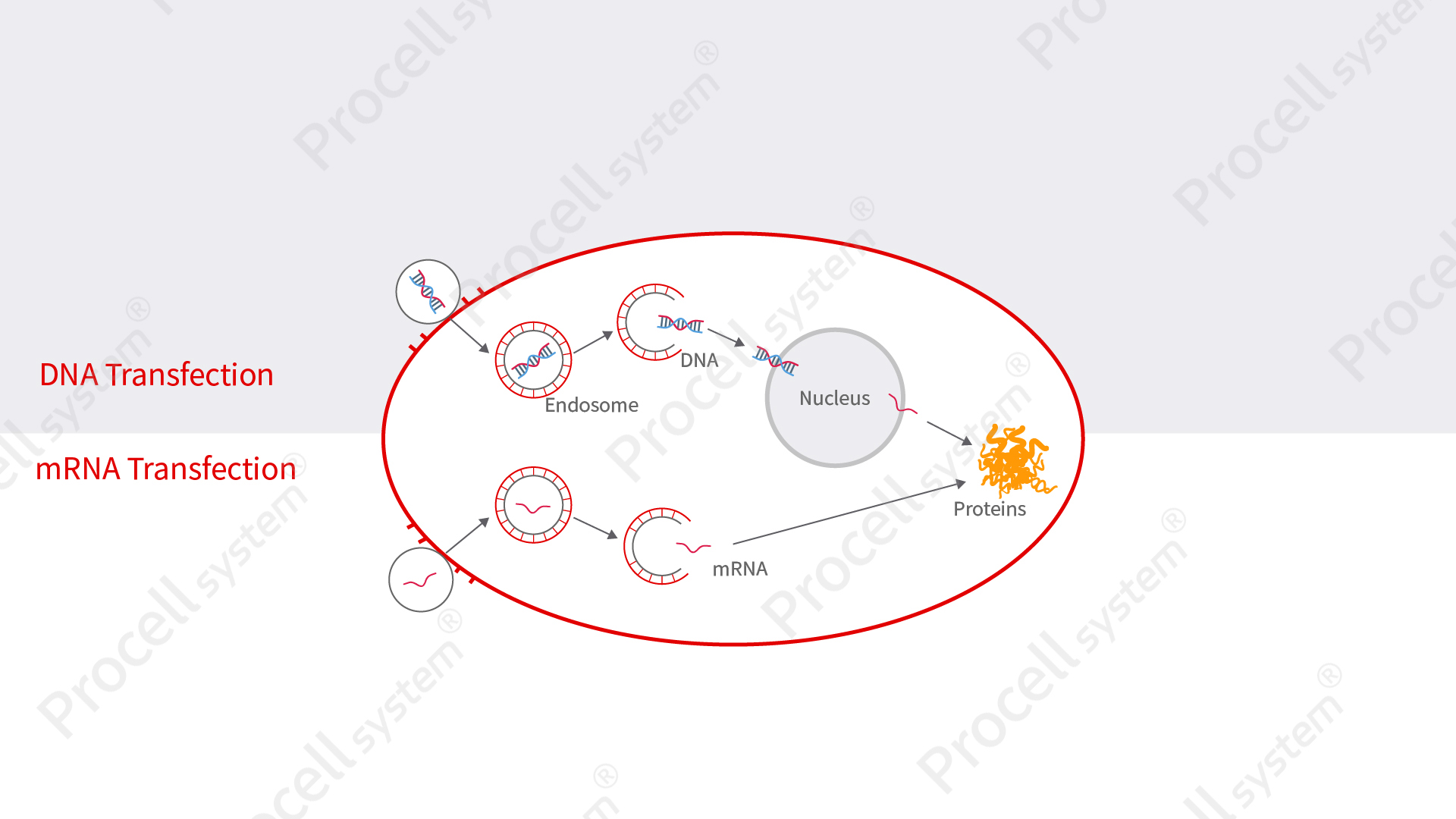
Figure 1. Schematic Illustration of the Principles of DNA and mRNA Transfection
Ⅱ. Why Does mRNA Transfection Often Fail?
Achieving robust and reproducible mRNA expression requires a clear understanding of the entire trajectory from extracellular delivery to functional protein production. Even a well-designed mRNA construct must overcome multiple biological barriers to be successfully translated.
1. Intrinsic Instability of mRNA
mRNA is a single-stranded RNA molecule whose phosphodiester backbone is highly susceptible to degradation by ubiquitous RNases. It is therefore inherently unstable, both during in vitro handling and within the intracellular environment.
In addition, unmodified mRNA can be recognized by innate immune sensors, such as Toll-like receptors and RIG-I-like receptors, triggering inflammatory signaling and interferon responses. This immune activation not only suppresses target protein translation but may also induce significant cytotoxicity.
2. Delivery Barriers
mRNA is a negatively charged, hydrophilic macromolecule that cannot freely diffuse across the lipid bilayer of the cell membrane. Efficient intracellular delivery requires encapsulation within an appropriate carrier, followed by cellular uptake, typically via endocytosis.
Once internalized, the mRNA-carrier complex must escape from endosomes into the cytoplasm in a timely manner. Failure to achieve endosomal escape results in trafficking through the trafficking through the endolysosomal pathway, leading to progressive degradation and loss of functional expression.
Therefore, optimization of mRNA transfection must address the entire workflow, including molecular design such as stability and immunogenicity, carrier selection and formulation such as cellular uptake and endosomal escape, and cell preparation and culture conditions to establish a permissive transfection environment. Limitations at any stage can significantly compromise overall expression efficiency.
Ⅲ. Key Parameters for Optimizing mRNA Transfection
Successful mRNA transfection results from the coordinated optimization of multiple variables. Systematic optimization is generally recommended across the following four parameters.
1. Cell Status and Culture Conditions
Use healthy cells at low passage numbers and confirm they are free of mycoplasma and other contamination.
At the time of transfection, cell confluence should typically be maintained at 70-90%, with adjustments based on the manufacturer's recommendations. Both insufficient and excessive cell density can compromise transfection efficiency.
2. mRNA Quality Control
mRNA generated by in vitro transcription must undergo DNase I treatment to remove residual template DNA, followed by purification using lithium chloride precipitation or silica column-based methods.
Ensure high RNA integrity, with an RNA integrity number preferably > 8.0, an OD260/280 ratio between 1.8 and 2.1, and no evident degradation bands.
3. Selection of Transfection Reagents
Choose commercially available transfection reagents that provide high delivery efficiency, low cytotoxicity, and good reproducibility.
Because transfection performance varies across cell types, reagents optimized for specific cell types should be prioritized when available.
4. Transfection Procedure Optimization
Follow the manufacturer's recommended mRNA-to-reagent ratio as a starting point and further optimize based on the specific experimental system. Importantly, there is no universal set of optimal conditions for mRNA transfection. Optimal performance can only be achieved through systematic evaluation and refinement to identify the most suitable combination of cell type, mRNA construct, experimental objective, and transfection reagent.
IV. Evaluation of Transfection Efficiency
The period 24-48 h post-transfection is critical for assessing transfection efficiency.
1. Immunofluorescence: When mRNA encoding a fluorescent reporter (e.g., EGFP) is used, the proportion of positive cells and the intensity of fluorescence can be directly visualized under a fluorescence microscope.
2. Flow Cytometry: Flow cytometry enables precise quantitative analysis of transfection efficiency, providing both the percentage of positive cells and the mean fluorescence intensity (MFI).
3. Functional Assays:
Western blotting can be used to assess the expression level of the target protein.
ELISA allows quantitative measurement of secreted proteins.
Luciferase reporter systems (e.g., MetLuc) can be employed to quantify reporter gene expression activity.
V. Frequently Asked Questions
Q: Why is mRNA unstable, and how should it be handled?
A: mRNA is highly susceptible to RNase-mediated degradation. RNase-free reagents and consumables (e.g., pipette tips, microcentrifuge tubes, and solutions) must be used throughout the procedure to prevent contamination. Aliquoted mRNA should be stored at −80°C for long-term storage, and repeated freeze-thaw cycles should be avoided. During preparation and handling of working solutions, samples should be kept on ice to maintain mRNA stability and activity.
Q: What causes inconsistent transfection efficiency?
A: Cell passage number should be strictly controlled, and healthy cells in the log phase should be used. The purity and quality of the mRNA must also be ensured. Variability is commonly associated with heterogeneous cell conditions or suboptimal mRNA quality.
Q: What can be done if cytotoxicity is excessive?
A: Shorten the incubation time between the mRNA-carrier complexes and cells (e.g., replace the medium after 4-6 h) to reduce toxicity from prolonged exposure. Additionally, decreasing the mRNA dose or optimizing the carrier-to-mRNA ratio may help mitigate cytotoxic effects.
Q: How can immunogenicity be minimized?
A: For applications sensitive to immune activation (e.g., stem cell studies or in vivo experiments), the use of high-purity, HPLC-purified mRNA is recommended to reduce immunostimulatory responses.
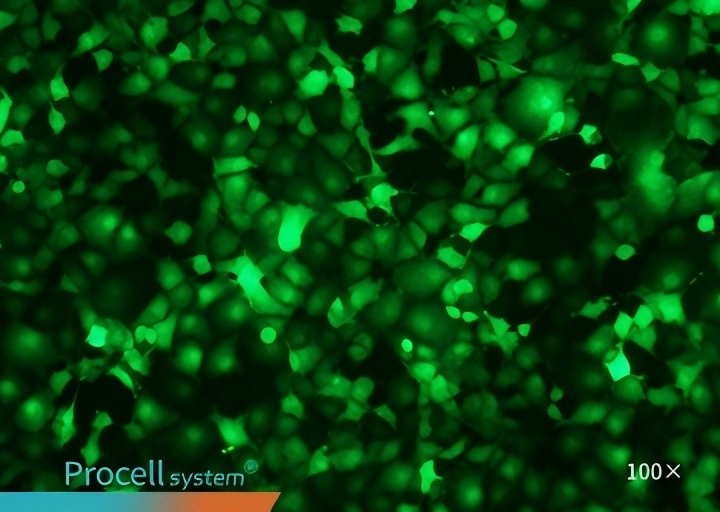
Selecting a cell type-matched transfection strategy is critical for maximizing mRNA transfection success. For example, cell-matched Procell Mergene1000® Cell-Specific mRNA Transfection Reagent outperforms T Brand L3000 in EGFP-mRNA delivery.
| Mergene1000® | T Brand L3000 | |
| CHO-K1 | 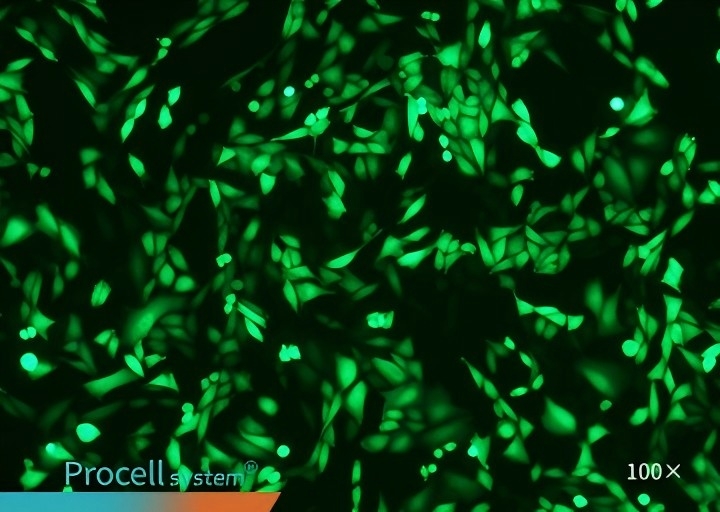 |
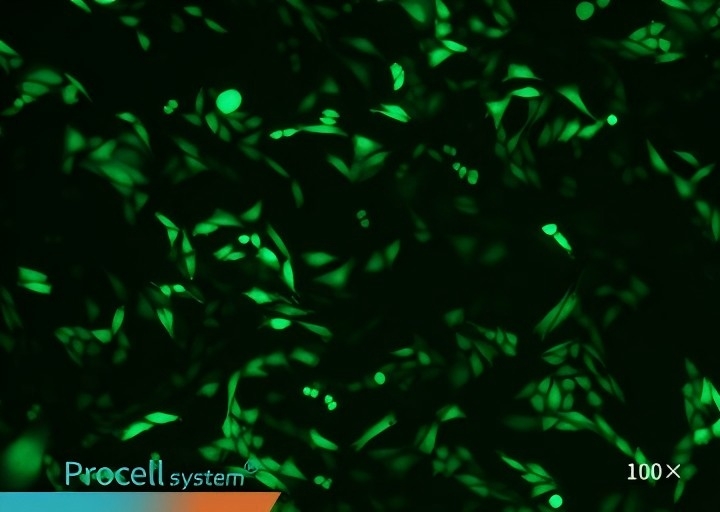 |
| Vero | 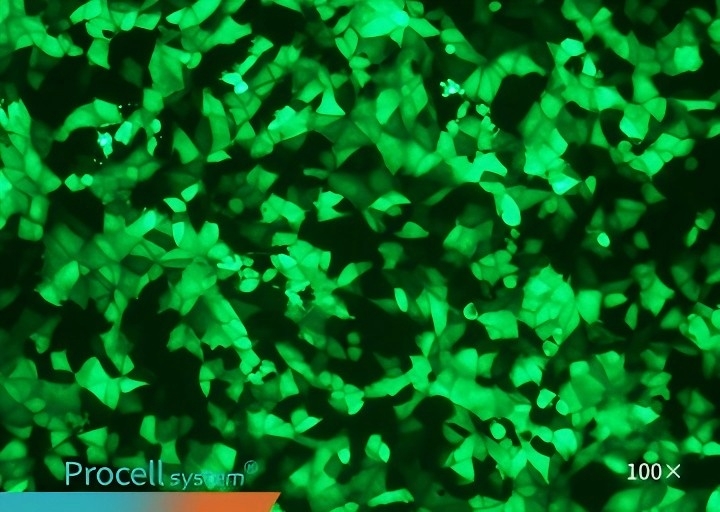 |
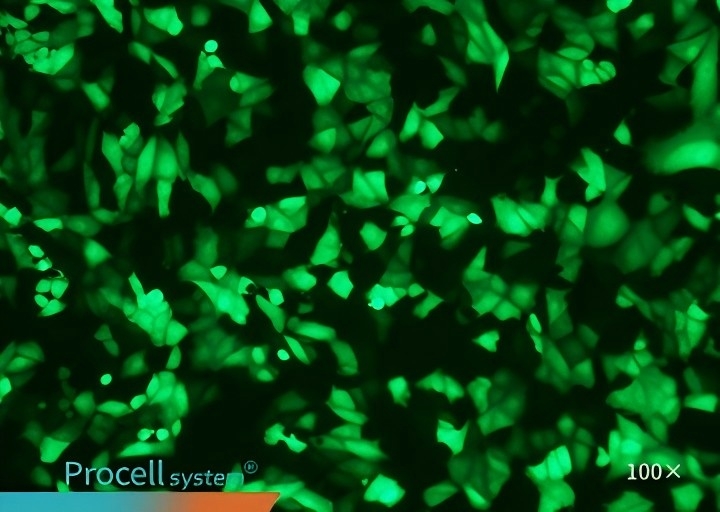 |
| COS-7 | 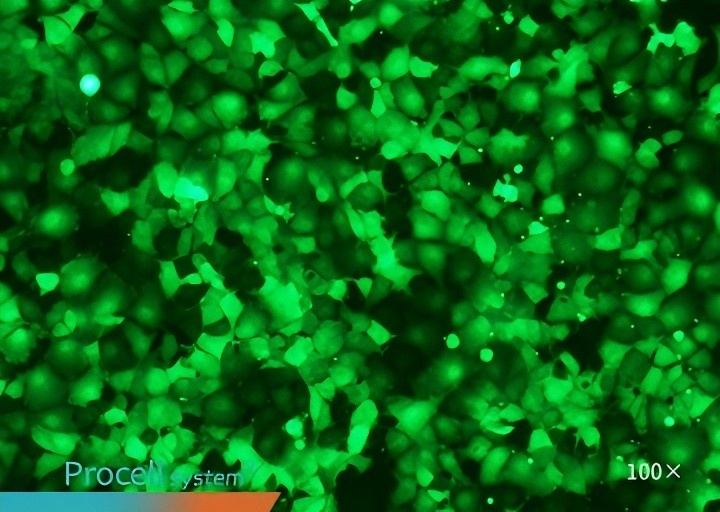 |
 |
For more cell-specific mRNA transfection reagents, please click Transfection Reagents to view the full product portfolio.
Prev: Cell Cryopreservation Guide: Serum-containing vs. Serum-free Freezing Media


