Cell Cryopreservation Guide: Serum-containing vs. Serum-free Freezing Media
Feb 12,2026
In cell culture, cells must be cryopreserved for short-or long-term storage when they are not actively cultured. However, although the freezing procedure is straightforward, post-thaw recovery is often suboptimal.
Why can cells withstand freezing? What factors lead to cryopreservation failure? And which methods are appropriate for different applications?
In this issue of Cell Culture Academy, we summarize the principles and practical strategies of cell cryopreservation, with emphasis on the rationale for selecting cryopreservation media to support informed experimental decisions.
I. Principles of Cell Cryopreservation
Cell cryopreservation aims to suppress cellular metabolic activity at low temperatures while minimizing structural and functional damage during freezing and thawing. This protection depends on the combined effects of low temperature and cryoprotective agents (CPAs).
The primary functions of CPAs include:
1.Rapid penetration into cells to lower the freezing point and reduce intracellular ice crystal formation.
2.Increased membrane water permeability, promoting cellular dehydration and limiting intracellular ice formation.
3.Facilitation of water re-entry during thawing to mitigate osmotic swelling.
4.Dilution of electrolytes in unfrozen intra-and extracellular solutions to reduce osmotic and chemical injury.
Ⅱ. Pre-cryopreservation Considerations
Before cryopreservation, the following factors should be carefully evaluated:
1. Cell Condition
Assess cell density, viability, and morphology prior to freezing.
Viability should generally exceed 90% to ensure optimal post-thaw recovery.
Do not freeze senescent, contaminated, or stressed cells.
2. Selection of Cryopreservation Medium
The most commonly used permeating cryoprotectant is DMSO (dimethyl sulfoxide). A typical formulation contains DMSO, fetal bovine serum (FBS), and basal medium.
Because DMSO is cytotoxic at room temperature, particularly to sensitive cells such as hybridomas, its concentration must be optimized by cell type, typically 5-15%.
For serum-free cultures, serum-free cryopreservation media should be used.
3. Experimental Conditions and Operational Constraints
Cooling rate is critical for cell survival.
Depending on laboratory equipment, cells may be frozen using controlled-rate (programmed) cooling or direct placement at -80℃.
Different cell types vary in tolerance to each method, and the approach should be selected accordingly.
Ⅲ. Serum-containing vs. Serum-free Cryopreservation Protocols
Cryopreservation approaches are broadly classified as serum-containing or serum-free based on medium composition. Neither is universally superior; selection depends on cell type, experimental objectives, and operational conditions.
1. Serum-containing Cryopreservation
Serum-containing protocols typically require slow cooling. Common approaches include:
A.Manual stepwise cooling:
Cells are sequentially transferred through 4℃, -20℃, and -80℃ before storage in liquid nitrogen, approximating slow freezing. This method is useful when controlled-rate devices are unavailable but is operator-dependent and less reproducible; pilot testing is recommended.
B. Programmed cooling freezing container
Cryovials are placed in a freezing container at -80℃ overnight and then transferred to liquid nitrogen for long-term storage. This is the most widely used laboratory method. Some commercial containers require isopropanol, and all should equilibrate to room temperature before use.
C.Programmable freezer:
Cells are cooled from room temperature to below -80℃ at approximately -1 to -3℃/min before liquid nitrogen storage. This approach provides high accuracy and reproducibility and is preferred for cells requiring stringent preservation quality, though equipment and operating costs are higher.
2. Serum-Free Cryopreservation
With the expansion of regenerative medicine and cell therapy, concerns related to contamination risk, immunogenicity, and batch variability in serum-containing media have increased demand for serum-free alternatives. These systems typically:
Contain no animal-derived components, reducing ethical and biosafety risks.
Have chemically defined compositions with minimal batch variation.
Allow direct freezing at -80℃ after resuspension, without controlled-rate procedures.
May be formulated without DMSO for DMSO-sensitive cells.
Notably, non-controlled freezing does not imply universal suitability. Pilot freeze–thaw validation remains necessary to assess post-thaw viability and cellular function.
IV. Cryopreservation Strategies for Different Cell Types
1. Cell Lines
Most adherent cell lines: Both serum-containing and serum-free media are suitable. Noncontrolled freezing media allow direct storage at -80℃ after aliquoting, offering convenience and good cost efficiency.
Suspension cell lines: Serum-containing media are generally recommended for stable post-thaw viability and growth. However, some lines (e.g., NK-92) also tolerate serum-free, noncontrolled freezing protocols.
Special cell lines: Difficult-to-recover cells such as SH-SY5Y, A7r5, and CTLL-2 may respond poorly to standard formulations. A 90% FBS + 10% DMSO mixture combined with slow cooling is recommended.
2. Primary Cells
Primary cells are fragile in vitro and highly sensitive to freezing, with occasional failure of post-thaw recovery. Cryopreservation decisions should consider differentiation status and passaging capacity.
Terminally differentiated cells that cannot be passaged, or fewer than three passages, are generally not recommended for freezing; experiments should begin immediately after receipt.
Cells capable of ≥3-5 passages may be cryopreserved after evaluation, preferably using commercial serum-free media designed for primary cells.
Immortalized primary cells retain primary phenotypes but have limited proliferative capacity and remain sensitive to freezing. Increase freezing volume to ensure sufficient viable cells after thawing, and replace medium 48 h post-recovery. Serum-free, noncontrolled freezing media designed for primary cells are preferred; alternatively, 90% FBS + 10% DMSO with gradual cooling may be used.
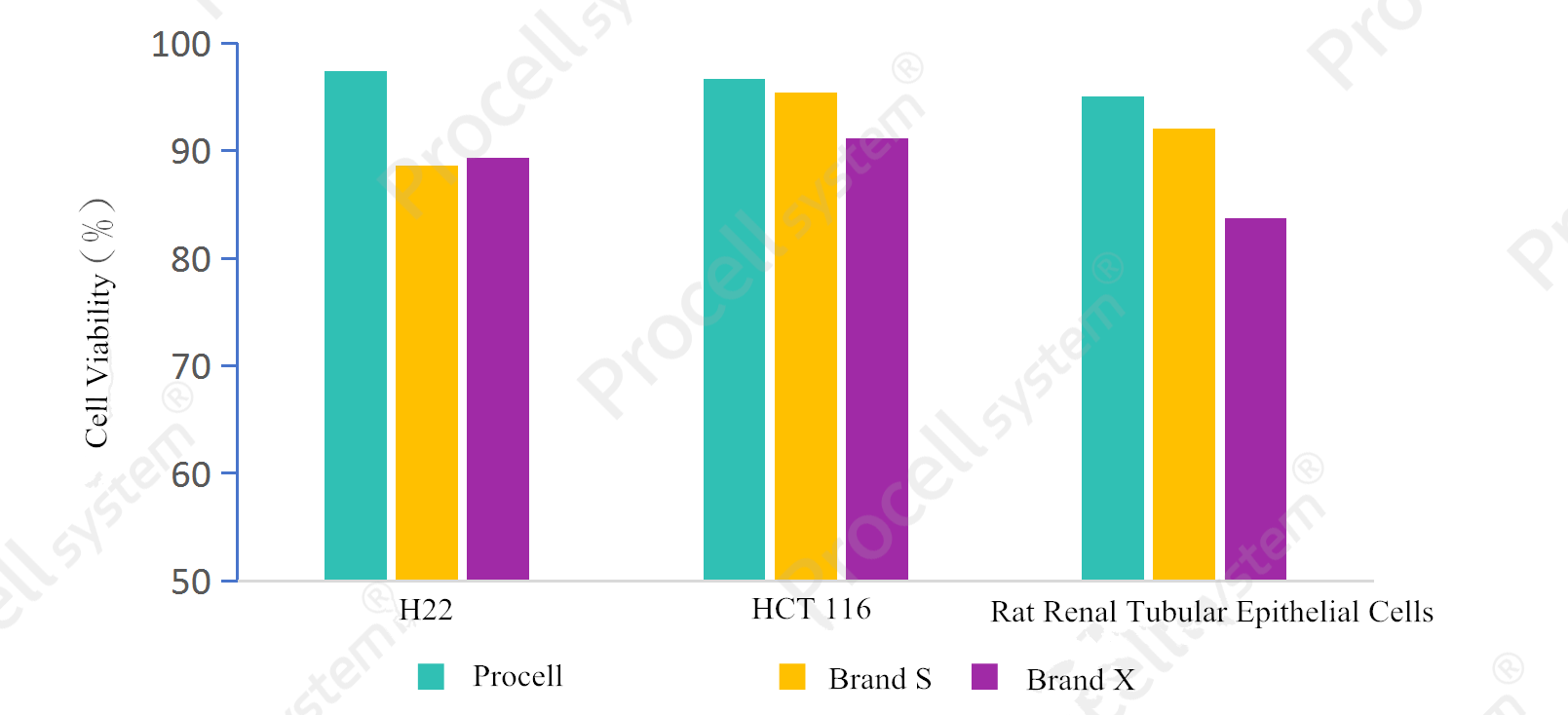
Procell Freezing Medium (Serum-free & animal origin-free) (Cat. No.: PB180438) supports cell cryopreservation with high post-thaw cell viability and a rapid fold increase within 48 h. It demonstrates consistent preservation performance across multiple cell types.
.png)
Prev: A Complete Guide to siRNA Transfection: From Cellular Entry to Precise Gene Silencing
Next: How to Boost mRNA Transfection Efficiency: Key Strategies Explained


