Why Do Angiogenesis Assays Fail? A Guide to Experimental Success
May 11,2026
Angiogenesis, the formation of new blood vessels from pre-existing vasculature, plays critical roles in wound healing, tumor progression, and metastasis. Aberrant angiogenesis contributes to the development of multiple diseases, including diabetic retinopathy and rheumatoid arthritis. Robust in vitro angiogenesis models are therefore essential for studying angiogenic mechanisms and screening therapeutic candidates. In this issue of Cell Culture Academy, we provide a practical overview of angiogenesis assays, including key principles, standard workflows, and common troubleshooting strategies.
I. Principles of Angiogenesis
Angiogenesis is a multistep process driven by endothelial cells and involves endothelial activation, migration, lumen formation, and vascular maturation[1].
1. Endothelial Cell Activation
Endothelial cells line the inner surface of blood vessels and possess migratory, proliferative, and extracellular matrix-remodeling capabilities. Angiogenesis is initiated when endothelial cells respond to external stimuli. Under hypoxic conditions, tissue injury, or within the tumor microenvironment, surrounding cells release growth factors such as VEGF. Binding of these factors to endothelial cell receptors activates downstream signaling pathways and induces endothelial activation.
2. Endothelial Cell Migration
Activated endothelial cells detach from existing vessels and migrate along growth factor gradients. During migration, they secrete matrix metalloproteinases (MMPs) to degrade the extracellular matrix while proliferating to support new vessel formation.
3. Tubular Structure Formation
Migrating endothelial cells subsequently align, connect, and undergo lumen formation to generate capillary-like tubular networks. This process requires coordinated cellular rearrangement and polarity establishment, while stable cell-cell junctions are essential for maintaining vascular integrity.
4. Vascular Maturation
Newly formed vessels are initially fragile and require further stabilization. Interactions among endothelial cells, pericytes, and smooth muscle cells strengthen the vascular wall, while integration with the existing vascular network establishes stable blood flow and functional vasculature.
In vitro angiogenesis assays exploit the ability of endothelial cells to retain angiogenic responsiveness and rapid migratory and proliferative capacities under culture conditions. In these assays, endothelial cells are seeded onto Matrigel, an extracellular matrix-rich substrate that mimics the basement membrane environment. Within hours, the cells migrate, reorganize, and form capillary-like networks.
Commonly used endothelial cell models include primary cells, immortalized primary cells, and established cell lines. Among these, human umbilical vein endothelial cells (HUVECs) are the most widely used due to their stable availability, strong responsiveness to VEGF, and ability to form reproducible, well-defined tubular networks in vitro[2].
Ⅱ. In Vitro Angiogenesis Assay
Successful angiogenesis assays require optimized experimental conditions and standardized procedures. Healthy HUVECs and a well-prepared Matrigel system are critical for reliable tubular network formation.
1. Experimental Design and Key Parameters
Before the assay, key variables such as medium volume, Matrigel usage, and cell seeding density should be optimized. Recommended parameters for different plate formats are listed below:
| Parameters | 96-well plate | 48-well plate | 24-well plate | 12-well plate | 6-well plate |
| Well growth area | 0.32 cm² | 0.95 cm² | 1.9 cm² | 3.8 cm² | 9.6 cm² |
| Matrigel volume | 50-75 μL | 150-200 μL | 200-250 μL | 300-400 μL | 600-800 μL |
| Cell seeding number | 25,000-30,000 | 50,000-75,000 | 75,000-150,000 | 150,000-250,000 | 300,000-500,000 |
| Medium volume | 100-125 μL | 250-300 μL | 400-500 μL | 800-1000 μL | 1.5-2 mL |
| Observation time | 4-6 h | 4-6 h | 4-8 h | 6-12 h | 6-18 h |
| Imaging difficulty | Moderate | Relatively easy | Easy | Easy | Easy |
Note: To reduce the risk of experimental failure due to suboptimal cell density, use a cell density gradient with multiple replicate wells.
2. Procedure (48-well plate example)
· Matrigel Coating
24 h before the experiment:
Thaw Matrigel slowly on ice at 4°C.
Pre-cool the 48-well plate and 200 μL pipette tips at 4°C. Pipette tip ends may be trimmed by 3–5 mm to improve handling of the viscous gel.
Keep all procedures on ice:
Gently mix the Matrigel with pre-cooled pipette tips and add 200 μL per well, avoiding bubble formation.
Note: Dispense the Matrigel slowly with the pipette tip held vertically above the center of the well to prevent adhesion to the side walls.
If the well bottom is not fully covered or the gel is unevenly distributed, gently tap or swirl the plate to ensure even coating.
· Gel Polymerization
Incubate the Matrigel-coated plate at 37°C for 45–60 min until fully polymerized.
Do not disturb the plate during polymerization to avoid uneven gel formation.
Inspect the gel surface during incubation. If ridges or grooves are observed, extend the incubation time until the surface is smooth before proceeding.
· Cell Preparation and Seeding
When HUVEC confluency reaches 70%–80%, detach cells with trypsin, centrifuge, and collect the cell pellet.
Resuspend cells in endothelial cell culture medium and count the cells.
Seed 6 × 104 cells per well in triplicate.
During seeding, hold the pipette tip vertically above the well without touching the gel layer, and add the cell suspension slowly to avoid disrupting the gel surface.
· Angiogenesis Incubation and Observation
Incubate cells at 37°C. Angiogenesis is typically well developed at approximately 4, when images can be collected for analysis.
0–1 h: Cells attach to the matrix. Do not disturb the plate or remove it from the incubator.
2–4 h: Tubular structures begin to form. During microscopic observation, keep the plate outside the incubator for no longer than 2 min.
>18 h: Tubular structures begin to degrade, and cells gradually undergo apoptosis and detachment.
Ⅲ. Common Issues and Troubleshooting
The Angiogenesis assay is highly sensitive to handling conditions. Common issues and recommended solutions are summarized below.
1. Uneven or “dune-like” gel surfaces result in nonuniform cell distribution.
Possible cause: This is typically caused by surface tension combined with the limited surface area of the well bottom.
Optimization strategies:
● Extend the Matrigel equilibration time to allow complete spreading and stabilization.
● Prefer 48-well or 24-well plates, as the larger surface area helps maintain a flatter gel surface.
Representative example:
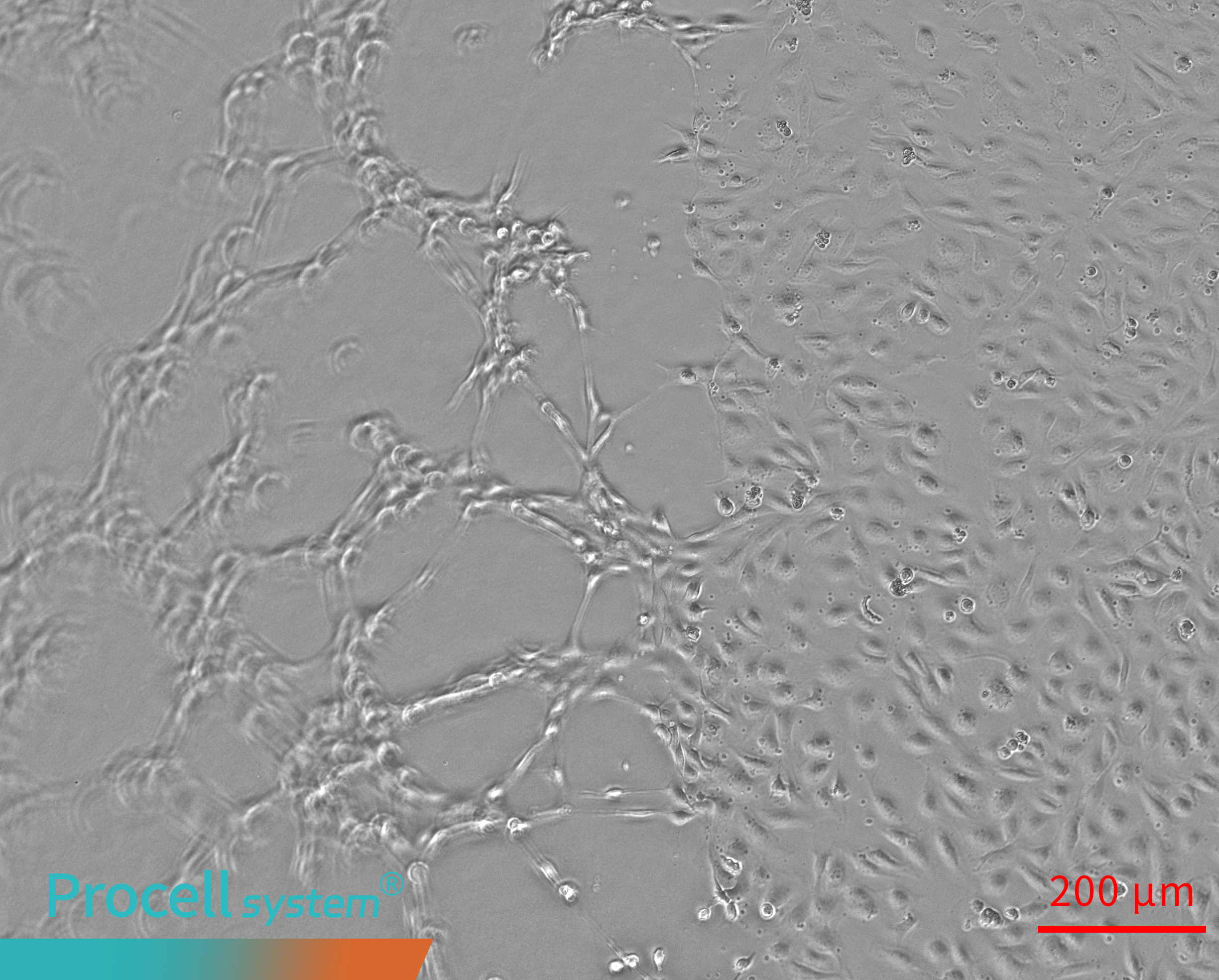
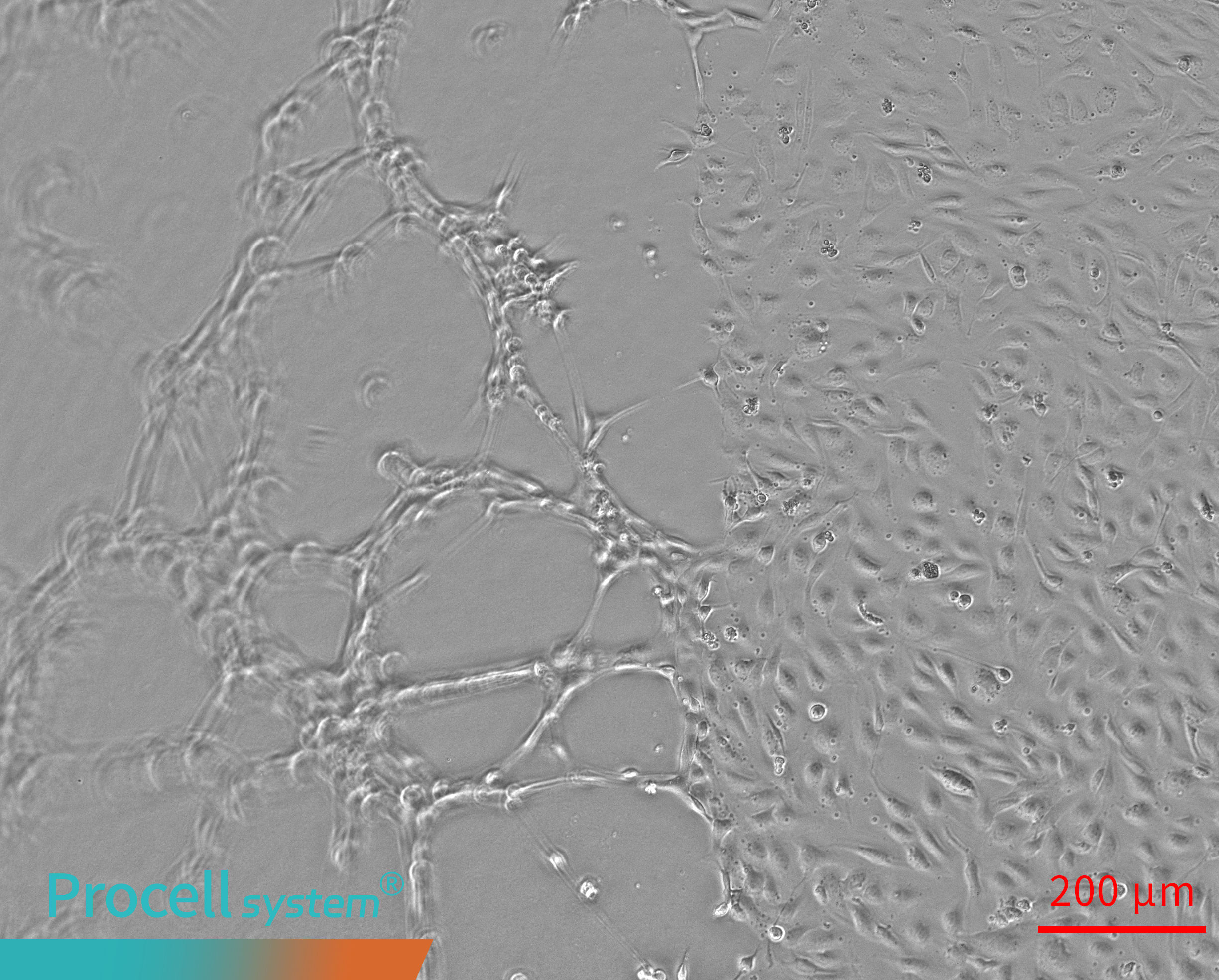
When the Matrigel surface is elevated at the edges and depressed in the center, Angiogenesis occurs mainly at the periphery, while network formation in the center is limited. In some cases, tubular structures may detach from the gel surface and appear suspended (Figure 1).
 |
 |
 |
 |
| Angiogenesis 0 h | Angiogenesis 2 h | Angiogenesis 4 h | Angiogenesis 6 h |
Figure 1. Uneven matrigel surfaces cause spatial bias in angiogenesis
2. Uneven Angiogenesis, with some regions lacking tubular structures.
Possible cause: This is usually caused by uneven cell seeding or nonuniform Matrigel distribution. Inappropriate cell density, inadequate mixing of the cell suspension, or improper dispensing can lead to localized cell aggregation or sparse distribution, disrupting cell–cell interactions and tube continuity.
Optimization strategies:
Optimize the seeding density and perform preliminary density gradient experiments if needed.
Thoroughly mix the cell suspension before seeding.
Hold the pipette tip vertically during dispensing to avoid disturbing the Matrigel surface.
Ensure even Matrigel coating without obvious thickness differences.
Representative example:
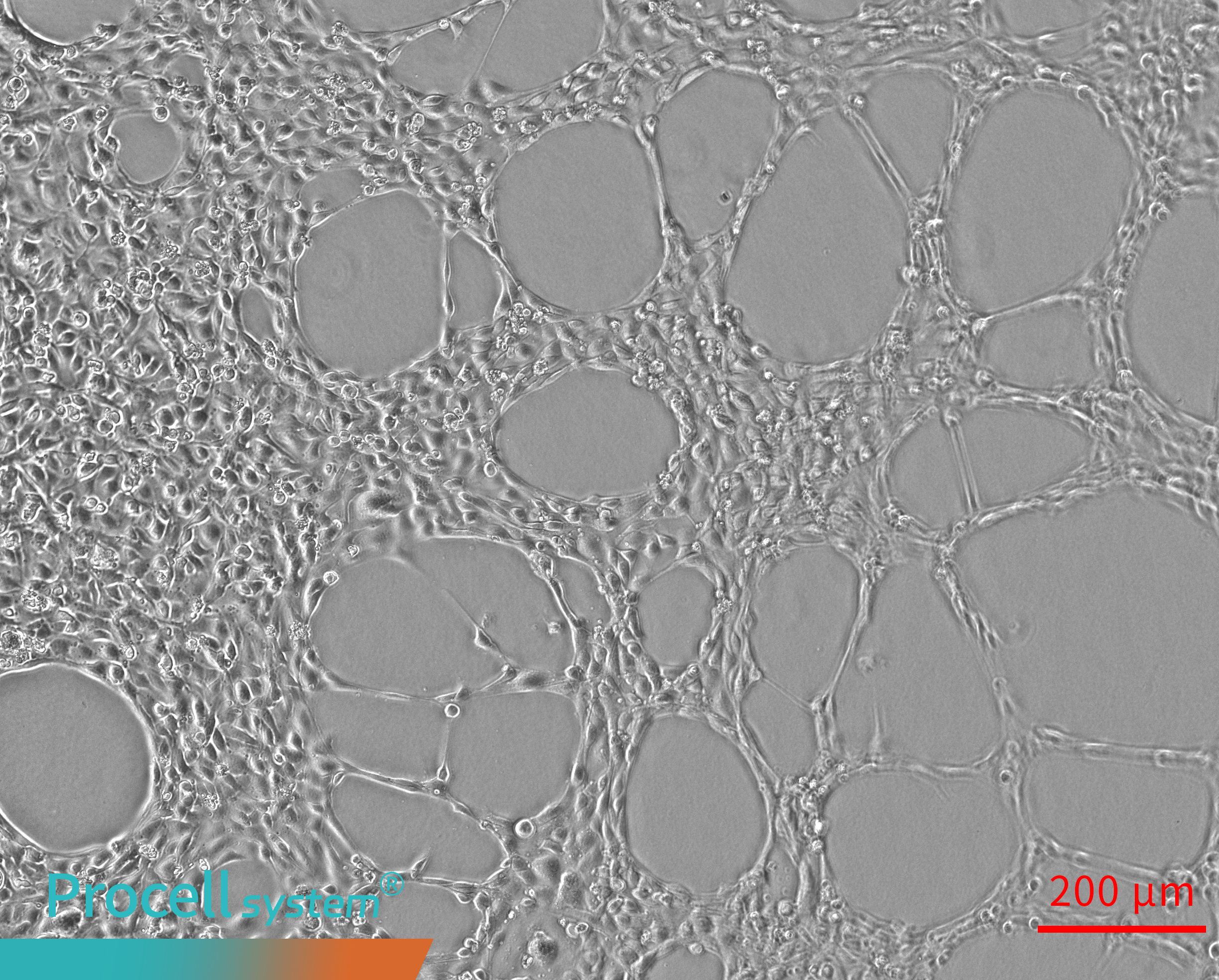
Uneven endothelial cell seeding leads to localized Angiogenesis over time; however, the overall network remains discontinuous, with some regions lacking tubular structures (Figure 2).
 |
 |
 |
 |
| Angiogenesis 0 h | Angiogenesis 2 h | Angiogenesis 4 h | Angiogenesis 6 h |
Figure 2. Uneven cell seeding results in discontinuous angiogenesis.
3. No obvious tubular networks are observed after 4 h of culture.
Possible cause: This is typically associated with poor cell quality or suboptimal culture conditions. Reduced viability, high passage number, low-quality or diluted Matrigel, or inappropriate culture medium can impair cell migration, intercellular connection, and branching.
Optimization strategies:
Use healthy, low-passage cells at 70%–80% confluency.
Verify the quality and concentration of the Matrigel, and avoid repeated freeze–thaw cycles.
Confirm that the culture medium contains endothelial cell growth factors.
Representative example:
Endothelial cells cultured on Matrigel for 0–6 h fail to form typical tubular networks. Cells remain dispersed or mildly aggregated, with limited migration, intercellular connection, and branching activity (Figure 3).
 |
 |
 |
 |
| Angiogenesis 0 h | Angiogenesis 2 h | Angiogenesis 4 h | Angiogenesis 6 h |
Figure 3. Insufficient Matrigel prevents tubular network formation.
Procell HUVEC complete media support robust and reproducible Angiogenesis on Matrigel.
 |
 |
| Human Umbilical Vein Endothelial (Primary cell immortalization) Cell Complete Medium | Human Umbilical Vein Endothelial Cell Complete Medium |
References
1.Jiang X, Wang J, Deng X, et al. The role of microenvironment in tumor angiogenesis. Journal of Experimental & Clinical Cancer Research. 2020; 39 (1):204.
2.Kelley M, Fierstein S, Purkey L, De Cicco-Skinner K. Endothelial cell Angiogenesis assay: an in vitro model for angiogenesis. Methods in Molecular Biology. 2022;2475:187-196.
Prev: Primary Cell Immortalization: Protocols for Culture, Cryopreservation, and Resuscitation


