Eliminate Cell Contamination: Key Types and Prevention Strategies at a Glance
Mar 20,2026
Contamination is one of the most persistent challenges in cell culture. Researchers at all levels have faced it: media turns turbid, cell morphology deteriorates, proliferation slows, and widespread cell death can occur—erasing days of work. Because contamination is difficult to eliminate once established, prevention is essential. Early detection, rapid assessment, and decisive action are key to minimizing losses.
In this issue of Cell Culture Academy, we review five common contamination types and highlight often-overlooked pitfalls to support fast identification and effective, evidence-based responses.
I. Bacterial Contamination
Bacterial contamination is the most common and fastest-spreading issue in cell culture, frequently involving Escherichia coli and Staphylococcus aureus.
Characteristics:
1. Microscopically visible as fine, dark, sand-like particles, with bacterial motility sometimes observed in some cases.
2. Typically cocci or rod-shaped, with uniform distribution.
3. Rapid growth leads to noticeable changes within 1 -2 d, including turbid media, pH shifts (phenol red color change), and occasional odor.
4. Overgrowth depletes nutrients and releases toxins, causing increased intracellular granularity, altered morphology, reduced proliferation, and potential large-scale cell death.
Prevention & Remediation:
1. Enforce strict aseptic technique and routinely clean the lab, incubators, and biosafety cabinets.
2. If contamination is observed, replace all related reagents and consumables immediately and trace the contamination source.
3. For low-level contamination, wash cells with high-dose penicillin-streptomycin Solution (e.g., 5× or 10× ), replace with fresh medium, and monitor closely.
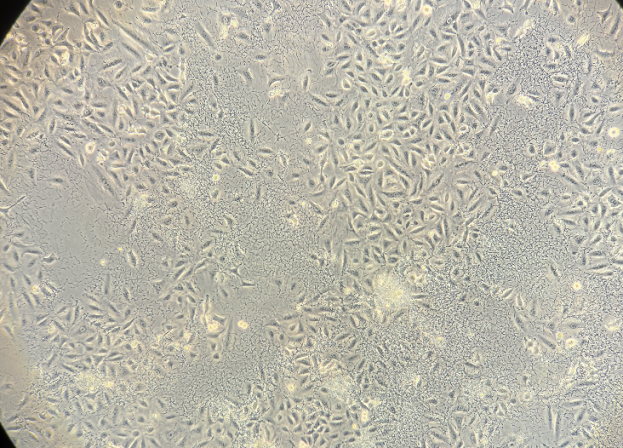 |
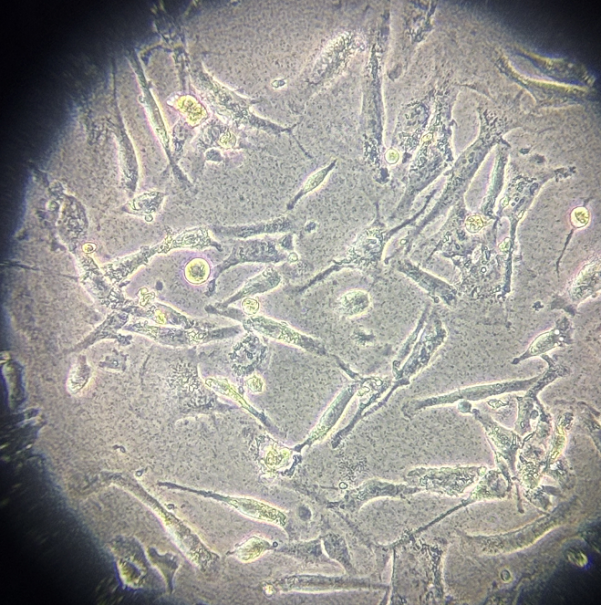 |
Figure 1. Representative morphology of cellular bacterial contamination (customer-reported case)
II. Fungal Contamination
Fungal contamination, primarily caused by molds and yeasts, spreads more slowly than bacterial contamination and has a longer latency period, but typically affects a broader area.
Characteristics:
1. Early-stage medium remains clear and are difficult to detect, despite localized cellular damage.
2. As contamination progresses, visible flocculent debris appears as white or yellow clumps in the medium. Microscopically, fungi present as chain-like, filamentous, tubular, or branching hyphae.
3. Airborne spores enable rapid cross-contamination, potentially spreading throughout the laboratory.
Prevention and Control:
1. Maintain relative humidity at 40 -60% and promptly remove waste to limit fungal growth.
2. Because fungal contamination is difficult to eradicate and highly transmissible, affected cells should be discarded after decontamination.
3. Thoroughly disinfect incubators, biosafety cabinets, and the laboratory to eliminate residual spores.
 |
 |
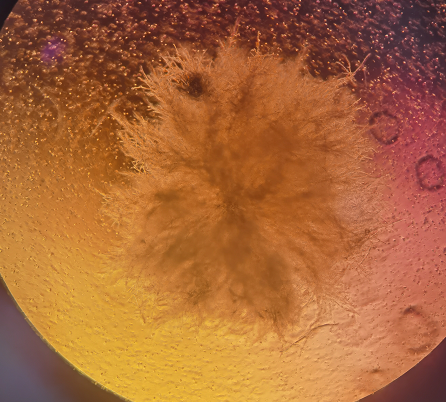 |
Figure 2. Representative morphology of cellular fungal contamination (customer-reported case)
III. Mycoplasma Contamination
Mycoplasma are the smallest self-replicating microorganisms, with a diameter of 0.1-0.3 μm, intermediate between bacteria and viruses. Undetectable by standard light microscopy, they are among the most elusive contaminants in cell culture.
Characteristics:
1. Irregular, non-motile dark particles may appear between cells.
2. Cells show declining cell health, morphological alterations (e.g., filamentous appearance), and reduced proliferation, eventually leading to detachment and cell death.
3.The medium typically remains clear and odorless.
Prevention and Control:
1. Routine screening is critical, using methods such as PCR, colorimetric assays, or fluorescence staining.
2. If the cell condition is acceptable, Anti-Mycoplasma Treatment Reagent may be used until test results are negative. Severely contaminated cultures should be decontaminated and discarded.
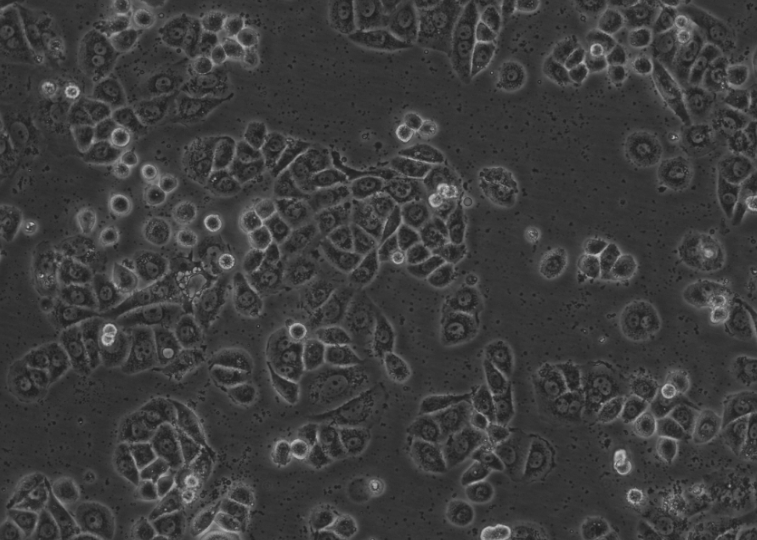
Figure 3. Representative morphology of cellular mycoplasma contamination (customer-reported case)
IV. Nanobacteria Contamination
Nanobacteria contamination is a poorly defined and controversial form of contamination, potentially associated with mycoplasma or other unidentified, low-proliferation microorganisms. It is difficult to detect by standard light microscopy.
Affected cells exhibit declining cell health and increasing numbers of small dark particles in intercellular spaces. These particles accumulate over time and cannot be removed by routine medium changes.
If the cell condition remains acceptable, Anti-Nanobacteria Treatment Reagent may be applied until cultures stabilize. If cell health is severely compromised, cultures should be decontaminated and discarded.
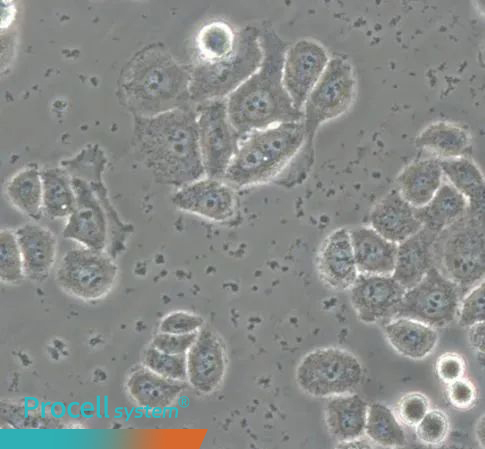
Figure 4. Representative morphology of cellular nanobacteria contamination
V. Cell Cross-Contamination
Cell cross-contamination is the unintended mixing of cells from different species or cell types within a single culture. Morphological changes may provide initial clues, but confirmation requires experimental validation.
Identification:
1. For cells of the same species, short tandem repeat (STR) profiling is recommended. Detection of more than three alleles at a locus may indicate cross-contamination, but results must be interpreted in the context of the cell’s genetic background (e.g.,EA.hy926 is a fusion cell line and naturally carries two sets of genetic information.).
2. For cells from different species, PCR amplification of species-specific genes enables identification based on differences in amplicon size.
If cell cross-contamination is confirmed, the culture should be re-established from an authenticated source.
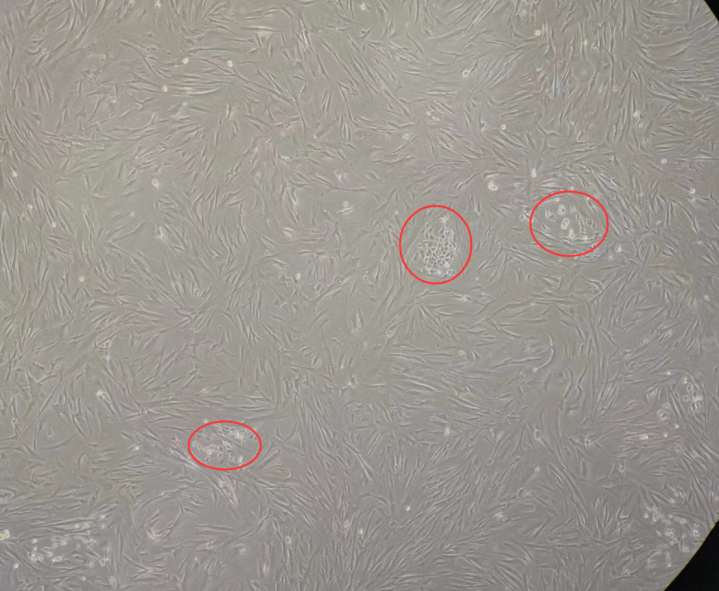
Figure 5. Representative morphology of cell cross-contamination (customer-reported case)
VI. Key Misconception: “Dark Spots” Do Not Always Indicate Contamination
The presence of “dark spots” under a microscope is often mistaken for contamination, but this is not necessarily the case. Similar features can arise from normal culture-related processes:
1. Cellular metabolic products: Secretory or metabolic granules released during normal cell growth. These are physiological byproducts and can usually be removed by medium exchange.
2. Cell debris: Mechanical stress during culture, such as incomplete enzymatic detachment followed by vigorous pipetting or tapping, can damage cells and generate fragmented debris.
3. Serum precipitation: Serum components (e.g., calcium phosphate, fibrin) may precipitate after improper storage or repeated freeze -thaw cycles, without affecting cell growth.
4. Apoptotic bodies: Changes in culture conditions may trigger apoptosis, producing apoptotic bodies. Stable culture conditions and minimizing reagent changes are recommended.
In summary, contamination should be determined by integrating cell growth status, morphological changes, culture conditions, and microscopic features, rather than relying solely on dark particles, to avoid misinterpretation and unnecessary concern.
Next: Primary Brain Cell Isolation: From Neurons to Blood-brain Barrier Cells


