Primary Brain Cell Isolation: From Neurons to Blood-brain Barrier Cells
Mar 24,2026
Primary brain cell isolation is a core technique for studying cellular behavior, signaling pathways, and disease mechanisms in the central nervous system. Yet, newcomers often face key questions: Why are 14-day-old animals used for some cell types, while 1-2-day-old neonates are preferred for others? Why do some protocols require density gradient separation, whereas others rely on direct enzymatic dissociation?
In this issue of Cell Culture Academy, we outline isolation strategies for five common primary brain cell types. By highlighting shared principles and critical differences, we provide a clear framework to support experimental design and optimization.
I. Characteristics of Five Major Primary Brain Cell Types
1. Brain Microvascular Endothelial Cells
Brain microvascular endothelial cells form a monolayer of flattened, cobblestone-like cells lining the microvascular lumen. They regulate substance exchange and vascular permeability, and also contribute to vascular tone, coagulation, inflammation, and angiogenesis. These cells are widely used in studies of thrombosis, atherosclerosis, inflammation, and tumor angiogenesis.
2. Brain Microvascular Pericytes
Pericytes are contractile cells that surround capillaries and venules and closely interact with endothelial cells. They regulate capillary blood flow, maintain blood-brain barrier integrity, and participate in angiogenesis, immune regulation, and clearance of metabolic waste. Pericyte dysfunction is a key focus in neuroscience research.
3. Cortical Neurons
Cortical neurons, composed of a cell body and processes, are the primary units of signal transmission in the brain. They are widely used in drug development and disease modeling.
4. Astrocytes
Astrocytes are the most abundant and relatively large glial cell types. In addition to structural support, they secrete diverse neuroactive molecules and are essential for maintaining the neuronal microenvironment, as well as regulating survival, migration, immune responses, signal transduction, axonal growth, and functional integration.
5. Neural Stem Cells
Neural stem cells are self-renewing, multipotent, with proliferative capacity. Through asymmetric division, they generate neurons, astrocytes, and oligodendrocytes, and are widely used in regenerative medicine and disease modeling.
II. General Workflow for Primary Cell Isolation
Primary brain cell isolation generally involves the following steps:
Tissue Collection: Dissect the target brain region under sterile conditions, keeping tissues on ice to preserve viability.
Tissue Dissociation: Mince tissue and apply appropriate enzymes, concentrations, and digestion times based on cell tolerance, balancing efficiency and viability.
Filtration: Pass the cell suspension through a 70-100 μm strainer to remove undigested fragments.
Cell Isolation and Enrichment (Optional): Depending on cell type, cells may be obtained by direct culture or require enrichment via density gradients, differential adhesion, or surface markers.
Cell Seeding and Culture: Seed purified cells in defined media with specific factors. Use selective conditions to enhance cell purity.
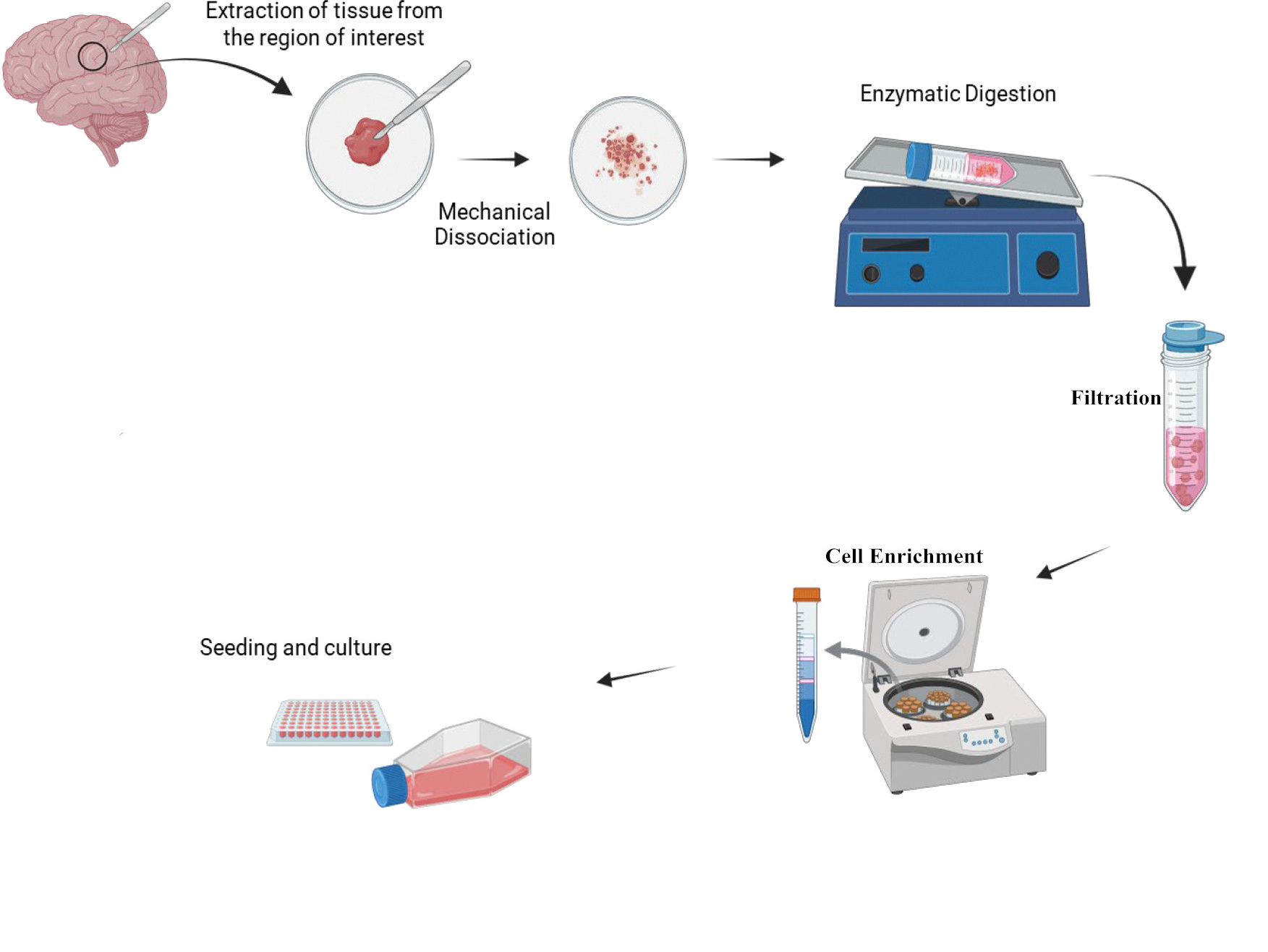
General Workflow for Primary Brain Cell Isolation (Source: online sources)
III. Key Differences in Isolating Distinct Brain Cell Types
1. Tissue processing
The cerebral cortex is a common source of multiple brain cell types, but preprocessing varies by target cell.
For vascular cells (brain microvascular endothelial cells and pericytes), visible surface vessels should be removed to avoid contamination from large vessel fragments.
For neural cells (cortical neurons, astrocytes, and neural stem cells), complete removal of the meninges is essential to prevent fibroblast contamination. Rapid dissection is also critical, as shorter processing times significantly improve cell viability.
2. Experimental Animals
Commonly used models include Kunming, C57BL/6, and BALB/c mice, as well as Wistar and Sprague-Dawley (SD) rats. Animal age is a key determinant of cell yield and viability.
| Cell Type | Recommended Age | Rationale |
| Brain Microvascular Endothelial Cells | 14-day-old | More mature vascular structures facilitate endothelial cell isolation |
| Brain Microvascular Pericytes | 14-day-old | Well-developed basement membrane and pericyte architecture support efficient isolation and purification |
| Cortical Neurons | 1-2-day-old | Softer tissue and reduced cell-cell adhesion improve neuronal survival after isolation |
| Astrocytes | 1-2-day-old | Higher glial abundance and strong proliferative and adhesive properties facilitate expansion and separation |
| Neural Stem Cells | 1-2-day-old | The neonatal subventricular zone contains a rich pool of neural stem cells |
3. Enzyme Selection for Tissue Dissociation
Vascular Cells: A two-step digestion protocol is used to release vascular fragments while minimizing cell damage.
Neural Cells: More sensitive to enzymatic digestion, these cells require carefully optimized conditions. Digestion should be promptly terminated to prevent overdigestion and preserve viability.
4. Isolation and Enrichment Differences
Vascular Cells: Density gradient centrifugation removes debris and myelin, enriching vascular cell populations.
Neural Cells: Generally do not require density gradients in routine protocols. Cells are obtained via mild enzymatic dissociation, with minimal mechanical stress to maintain viability.
5. Culture System Optimization
Primary isolates are typically mixed populations. Depending on cell type, purification or selective culture may be required to improve purity.
| Cell Type | Selection Required | Purpose |
| Brain Microvascular Endothelial Cells | Yes | Remove smooth muscle cells and pericytes |
| Brain Microvascular Pericytes | No | / |
| Cortical Neurons | Yes | Suppress glial proliferation |
| Astrocytes | No | Rapid adhesion and strong proliferation enable selective expansion |
| Neural Stem Cells | No | Suspension culture; no adhesion-based selection required |
Different cell types show distinct sensitivities to dissociation enzymes.
Accutase is recommended for passaging brain microvascular endothelial cells to preserve viability.
Brain microvascular pericytes can be passaged with trypsin.
Neural cells require milder dissociation and minimal mechanical trituration to reduce damage.
Prev: Eliminate Cell Contamination: Key Types and Prevention Strategies at a Glance
Next: A Practical Guide to Growth Supplements in Cell Culture for Stem Cells and Organoid Systems


