A Complete Guide to siRNA Transfection: From Cellular Entry to Precise Gene Silencing
Feb 10,2026
Small interfering RNA (siRNA) is a widely used tool for targeted gene silencing due to its high efficiency and sequence-specific mRNA degradation. In practice, however, unsuccessful siRNA transfection is more often caused by limitations of the transfection system, such as low delivery efficiency, cytotoxicity, and poor reproducibility, rather than by siRNA sequence design.
In this issue of Cell Culture Academy, we examine the full siRNA transfection workflow, from cellular uptake to precise cytoplasmic gene silencing. Emphasis is placed on delivery mechanisms, transfection optimization strategies, and critical aspects of result evaluation, providing practical guidance for achieving efficient, robust, and reproducible gene silencing.
I. The Delivery Challenge
Insufficient transfection efficiency and reduced cell viability are common causes of siRNA transfection failure. To function effectively, siRNA must overcome several key challenges:
1.Physical Barrier: The strong negative charge of siRNA prevents it from crossing the similarly charged plasma membrane unaided.
2.Biochemical Threats: Serum nucleases and intracellular lysosomes rapidly degrade naked siRNA, which has a half-life of less than 5 min under standard culture conditions.
3.Intracellular Traps: Even after uptake, most siRNA is sequestered in endosomes or degraded in lysosomes, preventing access to its site of action—the cytoplasm.
Transfection reagents are therefore essential to package and deliver siRNA. Any deviation, such as poor cell health, inappropriate reagent selection, or suboptimal handling, can compromise the experiment.
Ⅱ. The Nature of Transfection Reagents
Transfection reagents, typically cationic lipids or polymers, facilitate siRNA entry into cells through a “microscopic long march” across multiple cellular barriers:
1.Complex Formation: Positively charged reagents electrostatically bind negatively charged siRNA to form stable nanoscale complexes, protecting the nucleic acid and priming it for endocytosis.
2.Endocytosis: Complexes are internalized via clathrin-dependent or -independent pathways, forming endosomes. The nucleic acid remains encapsulated and protected at this stage.
3.Endosomal Escape: Escape from endosomes is the rate-limiting step. Complexes must reach the cytoplasm before endosome acidification and lysosomal fusion; otherwise, siRNA is degraded.
4.RISC Assembly: In the cytoplasm, the double-stranded siRNA unwinds, and the antisense strand is loaded into the RNA-induced silencing complex (RISC).
5.Target Cleavage: Guided by the antisense strand, RISC recognizes and cleaves complementary target mRNA, inducing degradation and achieving gene silencing.
III. Transfection System Optimization
The main challenge in siRNA transfection is maximizing the number of molecules that successfully escape endosomes while minimizing cytotoxicity. Optimization can target several key factors:
1.Cell Condition: Cells should be healthy and in logarithmic growth. For adherent cells, 50-70% confluency at transfection is recommended; for suspension cells, adjust according to log-phase growth. Too low or too high in cell density can reduce transfection efficiency.
2.Transfection Reagent Selection: The reagent is a critical determinant of delivery efficiency. Prioritize siRNA-specific reagents designed for the target cell type rather than generic reagents. Hard-to-transfect cells may require testing multiple reagents.
3.Reagent and siRNA Concentration: Transfection efficiency and cytotoxicity depend on reagent and siRNA amounts. Insufficient reagent lowers delivery, whereas excess increases cytotoxicity. High siRNA concentrations raise costs and may cause off-target effects. Optimize via gradient experiments; for example, in a 24-well plate, start with siRNA at 10-50 nM and adjust reagent volume to achieve the best balance between silencing and cell viability.
4.Incubation Time: Prolonged exposure to siRNA-reagent complexes increases cytotoxicity. For sensitive cells, replace the transfection medium with complete growth medium after 4-6 h to maintain silencing efficiency while improving cell viability.
IV. Outcome Assessment
1. Silencing Efficiency Validation
Fluorescently Labeled siRNA: During initial optimization, fluorescently labeled siRNA (e.g., FAM) enables rapid, visual assessment of cellular uptake via flow cytometry or fluorescence microscopy, providing an early measure of delivery efficiency without waiting for functional outcomes.
mRNA Level: Quantify target mRNA by qPCR 24-48 h post-transfection. Knockdown is typically maximal at this stage, though kinetics may vary with cell type and siRNA potency.
Protein Level (e.g., Western Blot): Protein reduction lags behind mRNA knockdown due to pre-existing protein turnover. Detection is usually performed 48-96 h post-transfection, depending on the target protein’s half-life.
2. Cytotoxicity Assessment
CCK-8: Evaluate cell viability 48 h after transfection. Viability ≥85% generally indicates minimal cytotoxicity.
Morphological observation: Cell morphology was assessed microscopically for apoptotic features, including cell shrinkage and detachment, providing complementary validation for CCK-8 assays.
V. Critical Details
1.Strict Control Setup: Include a positive control (e.g., siRNA targeting a housekeeping gene) to validate the transfection system and detection methods, and a negative control (e.g., scrambled siRNA) to rule out non-specific effects.
2.RNase Contamination: Work surfaces, pipette tips, and consumables can harbor RNases, and even trace amounts can degrade siRNA. Always use RNase-free certified materials and conduct experiments in a clean, controlled environment.
3.Cell Type Considerations: Transfection protocols differ between adherent and suspension cells. For suspension cells, collect and wash on the day of transfection to ensure consistency.
Procell Mergene1000® siRNA Transfection Reagent is tailored for specific cell types, offering high transfection efficiency and robust gene silencing.
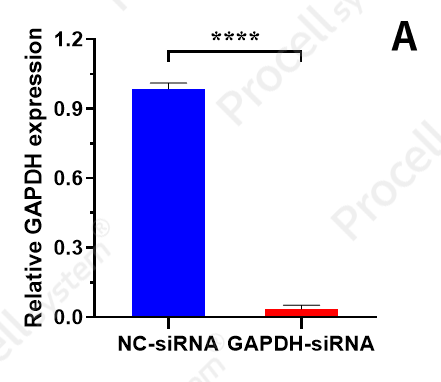 |
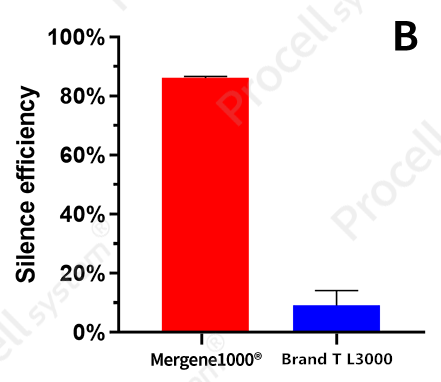 |
Mergene1000® NIH/3T3 Cell-Specific siRNA Transfection Reagent was used to deliver NC-siRNA and GAPDH-siRNA into NIH/3T3 cells and compared with Brand T L3000 (A: Relative GAPDH expression; B: Silencing efficiency).
Prev: Targeting the Root of Cancer: A Practical Guide to Cancer Stem Cell Research
Next: Cell Cryopreservation Guide: Serum-containing vs. Serum-free Freezing Media


