A Practical Guide to Growth Supplements in Cell Culture for Stem Cells and Organoid Systems
Mar 30,2026
The precision engineering of cellular microenvironments has become foundational to breakthroughs in regenerative medicine, disease modeling, and drug discovery. At the heart of this advancement lies the strategic application of growth supplements: defined formulations that deliver essential biochemical cues absent in basal media. These cell culture supplements critically influence cell survival, proliferation, differentiation, and functional maturation, particularly in sensitive models like stem cells and organoids. This guide explores the scientific rationale, classifications, and targeted applications of growth supplements, with dedicated analysis of two industry standard formulations: B-27 and insulin transferrin selenium (ITS). Emphasis is placed on evidence based selection to optimize experimental reproducibility and physiological relevance for researchers worldwide.
Table of Contents
1. Fundamentals and classification of growth supplements in cell culture
2. Auxiliary components enhancing cell culture stability and performance
3. Designing growth supplement strategies for organoid culture systems
4. Optimized use of growth supplements in stem cell research and expansion
1. Fundamentals and classification of growth supplements in cell culture
Growth supplements are precisely formulated additives incorporated into basal media to supply nutrients, signaling molecules, and protective agents necessary for robust in vitro culture. They are broadly categorized as serum based (e.g., fetal bovine serum), chemically defined serum free formulations, hormone supplements, and growth factor cocktails. Serum free options dominate contemporary research due to batch consistency, reduced immunogenicity, and regulatory compliance for clinical translation.
Specialized formulations consist of the B-27 supplement, initially created for neuronal cultures. The composition, known as b27 supplement components, includes antioxidants (vitamin E, catalase, superoxide dismutase), hormones (progesterone, putrescine), fatty acids, and trace elements. This multifaceted blend mitigates oxidative stress while supporting synaptic development and long term neuronal viability [1]. Conversely, the ITS supplement delivers insulin (functionally overlapping with igf 1 supplements), transferrin (iron transport), and selenium (glutathione peroxidase cofactor). ITS serves as a foundational cell culture media supplement for epithelial, mesenchymal, and progenitor cell systems where serum is contraindicated [2]. Classification further extends to application specific categories: growth factors in cell culture (e.g., EGF, FGF2, VEGF), hormone based additives (including growth hormone supplements), and niche mimetics tailored for organ specific lineages.
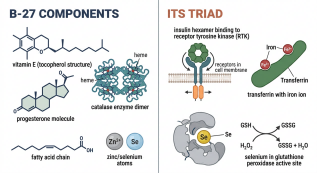
Fig. 1 Molecular architecture of core growth supplements.
2. Auxiliary components enhancing cell culture stability and performance
Auxiliary reagents encompass non nutritive yet indispensable components that stabilize culture conditions and potentiate primary supplement efficacy. While antibiotics, pH buffers (e.g., HEPES), and antifungals maintain sterility and homeostasis, certain auxiliary reagents function synergistically within growth supplement frameworks. The ITS formulation exemplifies this duality: insulin activates insulin and IGF 1 receptors to drive metabolic and proliferative pathways; transferrin ensures iron bioavailability; selenium neutralizes reactive oxygen species [3]. In mesenchymal stem cell growth medium formulations, ITS frequently replaces serum while preserving trilineage differentiation capacity [4].
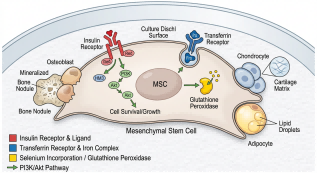
Fig. 2 ITS mechanism in mesenchymal stem cell maintenance.
Additional auxiliary elements include lipid carriers (e.g., cholesterol, phospholipids), trace minerals (zinc, copper), and extracellular matrix proteins (laminin, vitronectin). Notably, growth hormone supplements are increasingly evaluated for their role in modulating stem cell quiescence and metabolic activity, though concentration dependent effects necessitate rigorous optimization [5]. These reagents collectively enable fully defined, xeno free culture systems critical for Good Manufacturing Practice (GMP) compliant workflows and reducing experimental variability across laboratories.
3. Designing growth supplement strategies for organoid culture systems
Organoid culture media represent highly sophisticated applications of growth supplements designed to recapitulate tissue architecture, cellular diversity, and functional maturation in three dimensional structures. Successful protocols integrate stage specific supplement regimens. For cerebral organoids, Neurobasal medium supplemented with B-27 provides neuroprotective support essential for cortical layering, neuronal maturation, and reduced glial overgrowth [6]. Similarly, intestinal organoid systems rely on ITS or B-27 as basal support alongside niche specific factors (Wnt3a, R spondin 1, Noggin) to sustain Lgr5+ stem cell compartments [7].
In hepatic and renal organoid models, customized blends of growth factors in cell culture, such as hepatocyte growth factor (HGF) and fibroblast growth factor 10 (FGF10), are combined with ITS to enhance epithelial polarization and tubulogenesis [8]. Dynamic media transitions (e.g., reducing mitogens during maturation phases) underscore the temporal precision required in supplement deployment. Innovations continue toward fully synthetic organoid culture media, eliminating animal derived components to improve reproducibility and accelerate therapeutic adoption.
Procell Quick Overview of Popular Products:
Table 1. Reagents for Organoid Research
| Product Name | Cat. No. |
| B-27 neuron Supplement, without insulin, 50 × | PB180645 |
| B-27 neuron Supplement, without antioxidant, 50 × | PB180646 |
| B-27 neuron Supplement, with vitamin A, 50 × | PB180637 |
| B-27 neuron Supplement, without vitamin A, 50 × | PB180638 |
| N-2 Supplement (100 × ) | PB180641 |
| G-5 Supplement (100 × ) | PB180640 |
| Insulin-Transferrin-Selenium (ITS-G), 100 × | PB180429 |
| Insulin-Transferrin-Selenium-Ethanolamine (ITS-X), 100 × | PB180431 |
| Insulin-Transferrin-Selenium-Sodium Pyruvate (ITS-A), 100 × | PB180430 |
4. Optimized use of growth supplements in stem cell research and expansion
Stem cell research demands supplements that preserve pluripotency, direct lineage commitment, or expand progenitor pools without genetic drift. Human embryonic and induced pluripotent stem cells commonly utilize media containing basic fibroblast growth factor (bFGF) with ITS or knockout serum replacement [9]. For mesenchymal stem cell growth medium applications, ITS is a cornerstone component; its insulin fraction partially substitutes for dedicated igf 1 supplements, activating PI3K Akt pathways to enhance proliferation while maintaining differentiation potential [10]. Growth hormone supplements are explored in hematopoietic and neural stem cell contexts to modulate self renewal, though outcomes remain cell type and dose dependent.
The B-27 supplement is extensively validated in neural stem cell cultures, where its antioxidant profile counters metabolic stress during expansion. In cardiac differentiation protocols, modified B-27 formulations (without insulin) support cardiomyocyte maturation and electrophysiological function. As clinical translation advances, chemically defined supplements like B-27 and ITS offer critical advantages: lot to lot consistency, reduced pathogen risk, and alignment with regulatory standards for cell based therapies.
Procell Quick Overview of Popular Products:
Table 2. Reagents for Stem cell Research
| Product Name | Cat. No. |
| Y-27632 Dihydrochloride Solution (10 mM) | PB180609 |
| SB431542 Solution (100 mM) | PB180611 |
| Recombinant Trypsin Solution, without phenol red | PB180240 |
| Recombinant Trypsin Solution, with phenol red | PB180241 |
Growth supplements are indispensable enablers of modern in vitro biology. Formulations like B-27 and ITS demonstrate that rational design incorporating antioxidants, hormones, growth factors in cell culture, and trace elements can accurately reproduce physiological niches. Strategic selection of cell culture supplements, informed by cell type, application stage, and regulatory requirements, directly impacts data quality and translational potential. Ongoing innovation in supplement chemistry, coupled with multi omics validation, promises increasingly precise tools to empower global research in stem cell science and organoid technology.
References:
[1] Brewer GJ, Torricelli JR, Evege EK, Price PJ. Optimized survival of hippocampal neurons in B27 supplemented Neurobasal a new serum free medium combination. Journal of Neuroscience Research. 1993;35(5):567 576.
[2] Chen Y, Li X, Zhang Q, Wang L, Liu Y. Insulin transferrin selenium supplementation enhances proliferation of human mesenchymal stem cells. Stem Cell Research and Therapy. 2015;6:123.
[3] Le Roith D, Bondy C, Yakar S, Liu JL, Butler A. The somatomedin hypothesis 2001. Endocrine Reviews. 2001;22(1):53 74.
[4] Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315 317.
[5] Ismail PM, DeMayo FJ, Amato P, et al. Role of the growth hormone insulin like growth factor system in mammalian reproduction. Endocrinology. 2001;142(12):5115 5120.
[6] Lancaster MA, Knoblich JA. Organogenesis in a dish modeling development and disease using organoid technologies. Science. 2014;345(6194):1247125.
[7] Sato T, Vries RG, Snippert HJ, et al. Single Lgr5 stem cells build crypt villus structures in vitro without a mesenchymal niche. Nature. 2009;459(7244):262 265.
[8] Dutta D, Heo I, Clevers H. Disease modeling in stem cell derived 3D organoid systems. Trends in Molecular Medicine. 2017;23(5):393 410.
[9] Thomson JA, Itskovitz Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145 1147.
[10] Gage FH. Mammalian neural stem cells. Science. 2000;287(5457):1433 1438.
Prev: Primary Brain Cell Isolation: From Neurons to Blood-brain Barrier Cells


